
COLLABORATIVE WORKING
MSD supports and partners with the NHS and healthcare organisations in a number of different ways.
Through a combination of Collaborative Working (multi-organisation) and Joint Working (direct to NHS provider partnering) we pool skills, experience and resources for the development and implementation of patient-centred projects. Our ability to continue with our long-standing collaborative efforts pivots on the Department of Health and Social Care’s joint working mandate and enables us to discover and invent new ways, every day, to help patients, their families and their loved ones.
Select a project below to see more about how we partner in both oncology and vaccines or to find out more please contact us.
Read some of our Collaborative Working stories here.
MSD COLLABORATIONS
Oncology Collaborative Working
Bladder – Active Projects
Belfast Health and Social Care Trust Bladder Cancer Pathway Development Programme
Project Title
Belfast Health and Social Care Trust Bladder Cancer Pathway Development Programme
Organisations Involved
Belfast Health and Social Care Trust and MSD
Summary
There is an opportunity in Belfast Health and Social Care Trust to optimise the bladder cancer pathway to improve service quality, service efficiency, productivity and patient experience. The desired outcome of this project is to improve the efficiency of, or components of the bladder cancer pathway versus current baseline, including the achievement of the 62- and 31-day cancer waiting time targets that include time to surgery, radiotherapy and systemic anti-cancer therapy. The project is intended to run for approximately 12 months.
Background
The project is required to help address increasing pressure on the bladder cancer pathway, particularly in reducing waiting times for surgery and access to systemic anti-cancer therapy (SACT). Healthcare professionals, including consultants, nurses, and pharmacists, are currently overextended, with expectations of rising demand for services in the future.
Project Objectives
The project aims to establish a baseline understanding of the current timelines for each segment of the bladder cancer pathway through a comprehensive review involving multi-disciplinary teams. It seeks to identify inefficiencies that contribute to delays in patient transit along the pathway and produce a detailed map of the existing pathway along with a gap analysis. Belfast Health and Social Care Trust may use the outputs to address identified inefficiencies, potentially improving outcomes for bladder cancer patients and supporting the achievement of the 62- and 31-day cancer waiting time targets.
Benefits
Benefits to Patients
- An improved patient experience through the bladder cancer pathway in Belfast Health and Social Care Trust.
- Potentially quicker time to treatment through improved pathway efficiencies.
Benefits to NHS
- Smoother and quicker referral process through the bladder cancer pathway from urgent suspected cancer referral to first treatment.
- Potential improvement of the 62- and 31-day cancer waiting time targets.
- Potential efficiencies leading to more patients being treated in a faster time.
Benefits to MSD
- Better understanding of the patient journey through the bladder cancer
- Enhanced reputation of MSD through partnership work.
- As a pharmaceutical manufacturer of oncology medicines, an indirect result of an improved pathway may be that MSD see usage of their NICE reimbursed medicines.
Funding & Resources
This project is a shared contribution of time between Belfast Health and Social Care Trust and MSD. No funding is associated with this collaborative working project.
GB-NON-12053 | October 2025
Breast & Gynae – Active Projects
Bradford Breast Cancer Pathway Development Programme (PDP)
Project Title
Bradford Breast Cancer Pathway Development Programme (PDP)
Organisations involved
Bradford Teaching Hospitals NHS Foundation Trust
Summary
There is an opportunity in Bradford to optimise the breast cancer pathway in line with the national best practice timed breast pathway to improve the service quality, service efficiency, productivity, and patient experience. The desired outcome of this project is an improved breast cancer pathway aligned with the national best practice timed pathway and achievement of the breast cancer 28-day Faster Diagnostic Standard (FDS) and the 31- and 62-day Cancer Waiting Time (CWT) Targets. The project intends to run for approximately 12 months.
Background
A national best practice timed breast cancer pathway has been created with the intention of improving patient experience through promoting quality cancer care and ensuring all breast cancer patients receive optimal cancer care. The aim of this Pathway Development Programme is to improve the efficiency in the symptomatic breast cancer pathway from referral (day 0) to first assessment in the breast one-stop clinic (currently day 15 or day 16). The aim is to see these patients on day 10 or earlier.
Project Objectives
The primary objective of this project is the optimisation of the breast cancer pathway across Bradford, specifically contributing towards:
- An optimised breast pathway aligned to the national best practice timed breast cancer pathway, mapping it from referral to initial assessment
- Patients on the breast cancer pathway are seen by day ten or earlier, saving five days on the current pathway performance
- Improved patient experience of the breast cancer pathway
- Achievement of breast cancer 28-day Faster Diagnostic Standard, 31-day treatment target and 62-day referral to treatment CWTs, and the % of breaches are reduced
- Demand for each step of the pathway is understood, and sufficient capacity is in place to meet this
- Measurement of the outcomes of the project and disseminating these outcomes within six months of the project completion
Benefits
Benefits / Impact to Patients
- Improved patient experience of the breast cancer pathway
Benefits to the NHS Partner
- Achievement of the breast cancer 28-day FDS, 31-day treatment target and 62-day referral to treatment CWTs
- Earlier referral, diagnosis, and treatment of breast cancer patients, improving the chance of a successful treatment outcome
- Reduction in ad-hoc breast clinics
Benefits to MSD
- Enhanced reputation of MSD through partnership work
- As a pharmaceutical manufacturer of oncology medicines, an indirect result of an improved pathway may be that MSD see usage of their NICE approved medicines
Funding & Resources
This project is a shared contribution of time between Bradford Teaching Hospitals NHS Foundation Trust and MSD. No funding is associated with this project.
GB-NON-11521 | July 2025
NHS Lothian Endometrial Cancer Pathway Development Programme
Project Title
NHS Lothian Endometrial Cancer Pathway Development Programme
Organisations Involved
NHS Lothian and MSD
Summary
There is an opportunity in NHS Lothian to optimise the endometrial cancer pathway to improve service quality, service efficiency, productivity and patient experience. The desired outcome of this project is to enhance outcomes for endometrial cancer patients and meet the Endometrial Cancer Quality Performance Indicators. The project is intended to run for approximately 12 months.
Background
The project is required to help address increasing pressure on the endometrial cancer pathway, particularly in reducing waiting times for surgery and access to systemic anti-cancer therapy (SACT) and radiotherapy. Although there have been improvements in care quality and patient outcomes over the past decade, the 2024 Endometrial Cancer Quality Performance Indicators report for NHS Lothian indicates several areas which are still needing enhancement. Additionally, healthcare professionals, including consultants, nurses, and pharmacists, are currently overextended, with expectations of rising demand for services in the future.
Project Objectives
The objectives of the project include establishing a baseline understanding of the current timelines for each segment of the endometrial cancer pathway through a comprehensive review involving multi-disciplinary teams. It aims to identify inefficiencies that contribute to delays in patient transit along the pathway and produce a detailed map of the existing pathway along with a gap analysis, which NHS Lothian may use to address identified inefficiencies and potentially enhance outcomes for endometrial cancer patients and meet the Endometrial Cancer Quality Performance Indicators.
Benefits
Benefits to Patients
- An improved patient experience through the endometrial cancer pathway in NHS Lothian.
- Potentially quicker time to treatment through improved pathway efficiencies.
Benefits to NHS Lothian
- Smoother & quicker referral process through the endometrial cancer pathway from urgent suspected cancer referral to first treatment.
- Potential improvement in the efficiency of the pathway (or components of the pathway) versus Quality Improvement Indicators published in 2024.
- Potential efficiencies leading to more patients being treated in a faster time.
Benefits to MSD
- Better understanding of the patient journey through the endometrial cancer pathway.
- Enhanced reputation of MSD through partnership work.
- As a pharmaceutical manufacturer of oncology medicines, an indirect result of an improved pathway may be that MSD see more appropriate usage of their SMC reimbursed medicines.
Funding & Resources
This project is a shared contribution of time between NHS Lothian and MSD.
GB-NON-11353 | May 2025
Breast & Gynae – Completed Projects
Ovarian Cancer Maintenance Therapy Clinic
Project Title
Ovarian Cancer Maintenance Therapy Clinic
Organisations involved
MSD (UK) Ltd and The Christie NHS Foundation Trust
Summary
The project was set up as an eighteen-month pilot of a “one stop shop” dedicated Ovarian Cancer Maintenance Therapy (OCMT) Clinic at The Christie, with a proposed start date of February 2022. The service was nurse-led, supported by a pharmacist and with clinical expertise provided by an oncology consultant. The pilot provided a dedicated ovarian cancer maintenance therapy clinic aimed at all eligible ovarian cancer patients. Due to a delay in nurse recruitment, the start date was pushed to 1st of April 2023.
Background
The Christie manages patients from across Greater Manchester, with a population of 2.8 million, which is the largest cancer network in the UK. It also provides second opinions for patients nationally and there is a private patient service available through Hospital Corporation of America (HCA) Christie. The Christie treats ovarian cancer patients from across Greater Manchester, and at the time, patients on ovarian cancer maintenance therapies were managed in the medic-led outpatient clinic together with patients on active chemotherapy regimens.
The number of patients requiring this treatment is growing and there is a backlog of patients due to the COVID-19 pandemic. The clinic aimed to manage these patients within a dedicated service, alleviating the backlog of patients.
Project Support
Key Milestones
- Nurse recruitment: Advance Clinical Practitioner (ACP) was appointed in May 2023.
- Pharmacist and consultant support: The service is supported by four consultants and three pharmacists.
- Clinic set up: Dedicated Poly-ADP Ribose Polymerase inhibitor (PARPi) streams set up in each of the gynae clinics. Four clinics per week.
- Patient referral: Referral pathway established. All eligible patients are assessed through the OCMT clinics.
- Patient throughput: Following the commencement of the pilot, four OCMT clinics per week have been set up seeing on average 45 patients per week.
- Business case for future funding: Full funding for the post following completion of pilot has been secured. ACP has been appointed to the role.
- Clinical audit: an audit evaluating real world outcomes of ovarian cancer patients receiving maintenance therapy through the pilot has been completed.
- Electronic Patient-Reported Outcome Measures (ePROMs): Data is now collected for all patients that opt in. This is an online questionnaire used to collect direct reports from patients about their health, such as disease symptoms, treatment side effects and quality of life. Reminders are sent via text or email 3 days prior to the patient’s appointment. Further work is being carried out looking at uptake rate, impact on treatment delivery, outcomes and barriers to patient participation.
Benefits Realised
Benefits/ Impact to patients
- Dedicated service and point of contact: The OCMT clinic provided a dedicated service and point of contact for managing patient treatment, which was highly appreciated by patients.
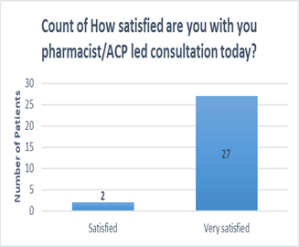
- High levels of satisfaction: The patient survey conducted over a 4-week period at the end of 2023 reported high levels of satisfaction with the service. 100% of patients felt satisfied with the appointments led by the ACP and pharmacists and felt confident in their ongoing monitoring1.
- Improved treatment options and compliance: The dedicated service improved treatment options and compliance, as evidenced by positive feedback from patients.
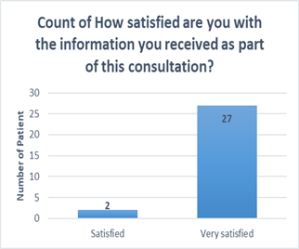
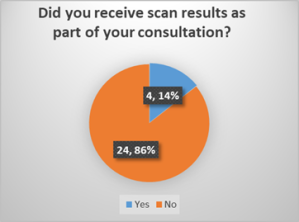
- Positive patient experience: All patients were satisfied or very satisfied with the explanation of the results and treatment plan.
- Reduced waiting time: Patients experienced less waiting time for their dedicated appointment slots at the OCMT clinic compared to a mixed clinic setting.
Benefits to the NHS partner
- More time for consultants: Patients managed within the nurse-led OCMT clinic resulted in consultants having more time available to deal with complex cases. Staff feedback indicated that the service took pressure off doctors in the clinic.
- Funding for a full-time NHS post has been secured.
- Service improvement: The service continuously improved by addressing areas of need or dissatisfaction, as highlighted by staff feedback.
- Provide other hospitals and services evidence to form business cases to adopt a similar service.
- Dedicated clinic to providing additional nurse time will aid with reducing post COVID backlog.
Benefits to MSD
- Lead Consultant willing to share experience of working with MSD to support future partnerships with wider NHS.
- Reputational benefit from partnering with The Christie.
- As a pharmaceutical manufacturer of oncology medicines, an indirect result of the ovarian cancer maintenance therapy clinic is that MSD may see appropriate use of their NICE approved medicines.
- This collaboration may have enabled more patients to access innovative treatments in line with NICE guidance which may or may not have included MSD medicines.
Patient Survey1
- A patient survey was conducted with 29 participants over a 4-week period at the end of 2023.
- High levels of satisfaction with the service reported.
- 100% of patients feel satisfied about the appointments led by the ACP and pharmacists and feel confident in their ongoing monitoring.
- All patients were satisfied or very satisfied with the explanation of the results and treatment plan.
Staff survey2
- Staff survey conducted November 2023. N=15
- 100% of staff feel the service has supported and improved the Gynaecology Medical Oncology outpatients’ clinics. High levels of confidence in the healthcare professionals who were leading the clinics.
Some comments from the survey:
“It takes the pressure off the doctors in clinic who are doing the mainstream patients.”
“We have a close integration between our pharmacists/ACPs and medics. It is this close relationship that supports the pharmacists/ACPs and which ensures that clinical care is maintained at the highest standards.”
“This has the potential to be a really brilliant addition to the gynae service – I think we just need to get our booking process right”
“The Lead Nurse and the Pharmacy team have made a tremendous impact on the OCMT service both in respect of providing a first of its kind high quality patient centred service and reducing the workload of the rest of the clinical team. The OCMT service is an excellent example of collaboration between the medical, nursing and pharmacy teams, and more widely between the Christie and Pharma industry.”
Stakeholder comments from the Sogolytic survey3:
“I am delighted with the success of our collaboration with MSD. I am hugely grateful to Wale Ajet and Nicky Harman for getting the project over the line. I have found working with other members from MSD very productive as well.”
Funding & Resources
This project was a shared funding commitment from The Christie & MSD. The total project cost was £103,130.
Lessons Learnt
- Incorporation of a pilot Nurse led clinic led to the project meeting its objectives.
- Having a Nurse Led clinic helped to release capacity of consultants.
- The importance of developing clinical guidelines and new pathways for clinic administration, treatment delivery and patient experience (ePROMS).
- This experience improved our understanding of collaborative working. A key takeaway for future projects is the importance of engaging with the Trust finance team at an earlier stage in the development process to ensure a more seamless integration of financial oversight.
Publications
There are no planned publications outside of this Outcomes Summary.
References:
- NHS Patient Survey Anonymised Raw data (data on file)
- NHS Staff Survey from The Christies May 2024 interim report (data on file)
- Sogolytic survey (data on file)
GB-NON-11112 | March 2025
Oxford TNBC Neo-adjuvant and Adjuvant Clinic Support
Project Title
Oxford TNBC Neo-adjuvant and Adjuvant Clinic Support
Organisations involved
Oxford University Hospitals NHS Foundation Trust (OUH) and MSD
Summary
The project’s goal was to redesign the treatment clinics for patients within the triple negative breast cancer (TNBC) service at the OUH. This enabled the service to better meet the growing patient volumes and demand on resources. To support this project, MSD provided funding of £24,000 which enabled the employment of a Pathway Coordinator. Further resources were pooled as MSD provided project management time, and the OUH provided Advanced Nurse Practitioner (ANP) project oversight for the service development initiative. The new Pathway Coordinator took on the administerial duties that the ANP team and Systemic Anti-Cancer Therapy (SACT) nurses would usually undertake. The duties included those which required a great deal of valuable specialist nursing time that otherwise could have been spent on patient treatment and care.
Employing a Pathway Coordinator enabled the ANP team to initiate and run clinics fully dedicated to the treatment and care of neo-adjuvant and adjuvant TNBC patients, as well as specialised SACT nurse clinic time for patients on oral SACT. Before we started the project, these patients were all seen in the same clinic where there can be a lack of focus and specific holistic care aimed towards those patients undergoing neo-adjuvant and adjuvant treatment. The project duration was 12 months, over which time we measured the capacity released in terms of CNS and ANP time freed up to focus on the running of new clinics to meet the growing patient demand.
Benefits Realised
This section describes how delivering this project has helped the TNBC service address the Trusts three quality domains: Patient Experience, Clinical Effectiveness and Patient Safety.
Patient Benefits – Neo-adjuvant and adjuvant SACT patients
- Dedicated clinic time now provides holistic and psychological support, addressing not only medical needs but also emotional and practical concerns. This includes managing side effects, adjusting prescriptions, and offering strategies to cope with treatment.
- Dedicated clinic time and greater focus may lead to fewer dose reductions and better management of side effects, which may help patients stay on treatment longer and improves their overall experience.
- Patients benefit from having a consistent point of contact who understands their treatment and can provide timely support.
Patient Benefits – Oral SACT patients
- Patients now experience improved care due to the increased availability of nurses for clinical duties.
- Service has moved from patients being given an 8-hour appointment window for telephone consultation, to patients now being given a 1 hour booking window, bringing the patient more convenient structure and timings.
- Patients now experience well-structured care and treatment planning, with better service coordinated and improved nurse organisation and engagement.
- Improved coordination of patient clinic bookings and blood appointments mean that the service has moved from booking just one appointment into the future, to now booking appointments for the next 4 months. It has been observed that this approach brings more certainty to a SACT patient at a time when there is a great deal of uncertainty.
NHS Benefits
Time and Cost Savings
- For the 5 months January to May 2024, the Pathway Coordinator has saved:
- 66 days (495 hours) of SACT nurse time.
- This equates to a B6 SACT nurse time cost equivalent saving of £3,678.
- When modelled out to 12 months, this would result in 158 days saved and a cost saving of £8,827.
- In addition, we can calculate that:
- £6.07 of nurse time equivalent is saved for every Band 3 Pathway Coordinator hour worked.
- 7 hours of SACT nurse time is saved per day for every day worked by Pathway Coordinator (of 7.5 hours).
Service Quality Improvement
- As a result of nurse time saved and capacity released, dedicated treatment clinic time has been created to focus on neo-adjuvant / adjuvant patients, with separate clinics being run for oral SACT patients. Dedicated treatment clinic time for neo-adjuvant and adjuvant patients has enabled:
- Patients now being seen at critical points in their treatment, such as after their first cycle of SACT, which is when they typically experience their first side effects. Timely management of side effects improves overall patient treatment experience.
- Anecdotally, the management of side effects early and effectively has reduced the need for patients to use triage services or be admitted to the hospital. This has resulted in cost savings and optimised the use of healthcare resources.
- Moving from clinic times not being harmonised and subsequent inability to engage junior doctors and consultants around treatment decisions, to now having all clinics running concurrently, enabling:
- Implementation of pre and post clinic meetings where senior clinical advice is sought in a timely manner.
- Improved clinical effectiveness where safer, timely and better-informed decision making takes place.
- Improved patient safety where treatment decisions are peer reviewed where needed, and made in a timely manner.
- Improved patient experience where patients don’t have to wait to find out what changes are needed to their treatment.
An increase in the number of patients seen in clinic
- Patient numbers seen in treatment clinic from Jan 2024 to May 2024 were observed to increase by 48% in 4 months (112 patients in January to 166 in May).
- This is due to SACT nurses now having a more clinically efficient service with better coordination of nurse resource and capacity that’s been released.
Clinical effectiveness
- We increased clinical compliance and effectiveness by ensuring that patients are put onto the correct nurses list relative to the stage they’re at in their treatment cycle.
- We reduced the amount of duplication and incorrect nurse attendances.
Nurse experience
- Anecdotally, job satisfaction and retention has improved as nurses are not having their clinical productivity reduced by admin duties.
- This is especially important given the OUH’s difficulties recruiting B6 SACT trained nurses, and the lengthy training requirements to upskill nurses to be SACT trained.
Recurrent funding for a substantive pathway coordinator role
- At the time of writing a Case of Need document (Business Case) has been written detailing the benefits of employing a Pathway Coordinator. This document has been used to secure continued ‘temporary’ funding while longer term, recurrent funding is still being secured. Despite current financial constraints within the NHS, the financial benefits, clinical efficiencies and improved patient experience from employing a pathway navigator have made it easier to justify the continued funding of the role in the near term. The benefits to the trust, the clinicians, the patient and the breast cancer service are clear and have been recognised by the senior OUH management team.
MSD Benefits
- Contributing towards the development of cancer services in the breast pathway, gaining experience of how to create service efficiencies, releasing capacity and improving patient experience.
- Collaborating on a project that will positively impact patients and has the potential to be scaled to other geographies as speaks to the NHSE cancer workforce agenda.
- This collaboration may have enabled more patients to access innovative treatments in line with NICE guidance which may or may not have included MSD medicines.
- Enhanced reputation of MSD through partnership work.
Funding & Resources
This project was a shared funding commitment from OUH & MSD. The total project cost was £24,000.
Lessons learnt
The project highlighted the importance of administrative support in improving clinical efficiency and patient care. Future collaborations should consider the value of dedicated administrative roles to alleviate the burden on clinical staff. Additionally, the project demonstrated the need for clear communication and collaboration between different departments to achieve the desired outcome.
Publications
None
GB-NON-10387 | January 2025
Head and Neck – Active Projects
Bradford Head and Neck Pathway Development Programme
Project Title
Bradford Head and Neck Pathway Development Programme
Organisations involved
Bradford Teaching Hospitals NHS Foundation Trust
Summary
There is an opportunity in Bradford Teaching Hospitals NHS Trust to optimise the head and neck cancer (H&N) pathway, in line with the national best practice timed head and neck pathway, to improve the service quality, service efficiency, productivity, and patient experience. This is to ensure patients receive timely and coordinated care throughout their cancer journey.
Background
A national best practice timed head and neck cancer pathway has been created with the intention of improving patient experience through promoting quality cancer care and ensuring all head and neck cancer patients receive optimal cancer care. The desired outcome of this project is an improved head and neck cancer pathway aligned with the national best practice timed pathway, and achievement of the head and neck 28 day Faster Diagnostic Standard (FDS) and the 31- and 62-day Cancer Waiting Time (CWT) targets. The project intends to run for approximately 12 months.
Project Objectives
The primary objective of this project is the optimisation of the head and neck cancer pathway. Specifically.
- Head and neck pathway is aligned to the national best practice timed
- Improved patient experience of the head and neck cancer pathway
- Achievement of 28 FDS, 31- and 62-days CWT
- % of CWT target breaches reduced
Benefits
Benefits/Impact to patients
- Improved patient experience of the H&N cancer pathway
Benefits to the NHS Partner
- Achievement of the H&N cancer 28-day FDS, 31-day treatment target and 62-day referral to treatment CWT
- Earlier diagnosis and treatment of H&N cancer patients, improving the chance of successful treatment outcome
- Reduction in did not attend (DNA) rate
Benefit to MSD
- Enhanced reputation of MSD through partnership work
- As a pharmaceutical manufacturer of oncology medicines, an indirect result of an improved pathway may be that MSD see usage of their NICE/SMC approved medicines
Funding & Resources
This project is a shared contribution of time between Bradford Teaching Hospitals NHS Foundation Trust and MSD.
GB-NON-12045 | November 2025
Head and Neck – Completed Projects
South Yorkshire and Bassetlaw Cancer Alliance Head & Neck Pathway Development Project (PDP)
Project Title
South Yorkshire and Bassetlaw Cancer Alliance Head & Neck Pathway Development Project (PDP)
Organisations involved
South Yorkshire and Bassetlaw Cancer Alliance: Sheffield Teaching NHS Foundation Trust, Barnsley Hospital NHS Foundation Trust, The Rotherham NHS Foundation Trust, Doncaster and Bassetlaw Teaching Hospitals NHS Foundation Trust and Chesterfield Royal Hospital NHS Foundation Trust & MSD-UK
Summary
A national head and neck cancer best practice timed pathway (BPTP) has been created with the intention of improving patient experience through promoting equality in cancer care and ensuring all head and neck cancer patients receive optimal cancer care. There was an opportunity in South Yorkshire to optimise the head and neck cancer pathways in line with the BPTP to improve the service quality, service efficiency, productivity and patient experience. Also, to improve cross-functional working relationships across all South Yorkshire and Bassetlaw Cancer Alliance Trusts including but not limited to ENT (ear, nose and throat) and OMFS (oral maxillofacial surgery) departments. The desired outcome of this project was an improved efficiency of the regional diagnostic head and neck cancer pathway aligned with the BPTP milestones, 28 day Faster Diagnostic Standard (FDS) and the 31- and 62-day Cancer Waiting Time (CWT) Targets. The primary objective was to implement the changes in the 5 foundation hospital trusts within the South Yorkshire Cancer Alliance region in partnership with Chesterfield. The project began on 1st January 2022 and finished on the 31st December 2022.
Benefits realised
Through implementation of MSD’s Pathway Development Programme with the South Yorkshire and Bassetlaw Cancer Alliance team, MSD provided project management support to assess the current state of the head and neck pathway and provided a gap analysis contrasting the pathway with the BPTP. MSD then supported the implementation of improvement initiatives to close these gaps and assisted with the measurement of their impact. Also, MSD facilitated the formulation of service improvement meetings within each of the 5 diagnostic hospitals. As a result of the project, the following benefits were realised:
Patient Benefits
- Quicker diagnosis and time to treatment of head and neck cancer has been reported through this project potentially leading to improved patient outcomes, See NHS benefits below:
- Patient experience was not measured
NHS Benefits
- Barnsley Hospital NHS Foundation Trust
- Referral of patient to when first seen by oncologist reduced from 9 days to 5 days
- Rotherham NHS Foundation Trust CT
- Reporting has reduced from 6 days to 4 days
- Sheffield Teaching Hospital NHS foundation Trust
- ENT referral of patient to when first seen by oncologist reduced from 10 days to 6 days
- ENT patients are now reviewed at MDT on day 35 vs day 40
- OMFS patients are now reviewed at MDT on day 36 vs day 40
- Doncaster and Bassetlaw Teaching Hospitals NHS Foundation Trust and Chesterfield Royal Hospital NHS Foundation Trust
- No data was collected
- Achievement of CWT & FDS was analysed from the 62- and 28-day CWT target performance data concluded the following:
- The 62-day CWT in January 2022 was 47.1% vs 52.9% in December 2022
- The 28-day FDS in January 2022 was 74% vs 77% in December 2022
- Data was an amalgamation of all the trusts within the South Yorkshire and Bassetlaw Cancer Alliance
- Treatment rates were not measured as part of this project
MSD Benefits
- Better understanding of head and neck cancer patient needs
- Enhanced reputation of MSD through partnership work. Four NHS stakeholders completed a survey following the PDP and the results are shown below:
- Average score of 8.75/10 were satisfied with MSD’s collaborative working project
- Average score of 8/10 believed MSD supported their organisations development or improved cancer service
- Average score of 3.25/10 to unlikely work with MSD and the pharmaceutical industry prior to the collaborative project which then changed to an average score of 8.25/10 in favour of collaboration following the project.
- Average score of 8.5/10 would recommend working in collaboration with MSD to others
- Quotes from NHS Stakeholders taken from the survey when questioned ‘what has improved in your service/pathway following this collaborative working project’:
- “Improvement in achievement of best practice timed pathway milestones, significant improvements in engagement with service improvement methodology and increased staff morale within the CDG leadership team”
- “Improved understanding of the pathway and barriers, data collection to inform developments, collaborative working through working groups and progress towards meeting BPTP (best practice timed pathway) milestones”
- “The involvement of MSD has resulted in vastly improved cohesion, consistency and efficiency across a number of aspects of the head and neck pathway in South Yorkshire & Bassetlaw”
- When prompted for ‘any feedback of your experience of working in collaboration with MSD’:
- “Wasn’t aware of the MSD collaboration projects prior to this”
- “Became a valued and welcome addition bringing expertise and previous experience”
- “Friendly and supportive in identifying the challenges that are faced in our area of work”
- This collaboration may have enabled more patients to access innovative treatments in line with NICE guidance which may or may not have included MSD medicines.
Funding & Resources
This was a project with shared time commitment from South Yorkshire and Bassetlaw Cancer Alliance, the participating Trusts & MSD.
Lessons learnt
- Importance of having access to tumour pathway steps timings data, and the challenges in extracting pathway timing data from Hospital IT systems. Recommendation – get agreement on data extract (both how and what) in project scoping phase.
- Wider NHS workforce challenges impacted on project delivery. Recommendation – engage project sponsors to ensure project work is prioritised throughout the duration of the project.
Publications
There are currently no plans to publish the outcomes of this project by the hospital trust as per the date of this summary.
Job Code: GB-NON-07849 | August 2023 Approved for continued use April 26
Community Pharmacy/General Practitioner (CP/GP) Head and Neck Cancer Pathway Project
Project Title
Community Pharmacy/General Practitioner (CP/GP) Head and Neck Cancer Pathway Project
Organisations involved
NHS Tayside
MSD-UK
Summary
NHS Tayside, through the North Cancer Alliance, worked in collaboration with MSD to develop a communication pathway between community pharmacies in the Dundee city area and their local primary care practices for patients with suspected head and neck cancers (HNC).
The project evaluated the effectiveness of a communication pathway between community pharmacy and primary care to determine whether it help to facilitate earlier patient review.
Project Objectives
The project would aim to deliver –
- A communication pathway between community pharmacies and primary care services in the Dundee city area aimed at identifying HNC’s
- A written evaluation of the feasibility of engaging community pharmacies to identify potential cases of HNC’s and the effectiveness of the communication pathway
- A road map for other Health Boards and/or Scottish Government to emulate/scale the project
Project Outcomes
The project aimed to recruit 20 pharmacies and conduct approximately 1000 consultations for suspected head and neck cancer symptoms. However, 14 pharmacies were recruited, with 9 completing the project and several hundred consultations were carried out for suspected head and neck cancer symptoms. The reasons for incomplete participation included pharmacists leaving, maternity leave, failure to undergo training, and disengagement with the project. The reasons for an estimated number of consultations is that all pharmacies aimed to record the consultations however due to not all pharmacies taking part and the continued requirement to use locum pharmacists, who may or may not been aware of the project, only an estimation can be made. The project identified two patients for the communication pathway.
Benefits Realised
Benefits/ Impact to patients
- The project has demonstrated that community pharmacy teams can be trained to recognise symptoms of head and neck cancer amongst people presenting at the pharmacy meaning that more patients may be identified earlier in their disease and potentially benefit from earlier treatment.
- The project has helped to expand the services offered to patients by community pharmacies, especially those in deprived areas where accessing health provision such as GP appointments is often difficult thus helping address health inequalities.
- Patients may feel more confident in initially going to a community pharmacy to discuss their symptoms knowing that staff have been upskilled and there is a communication pathway in place with their GP practice.
Benefits to the NHS
- The project has supported action points 16, 17 and 18 of the Scottish Cancer Action Plan and shows that community pharmacies can support earlier diagnosis of cancer and help a sustainable earlier cancer diagnosis solution.
- The training provided to the pharmacists has allowed pharmacists to be more confident in identifying the potential symptoms of head and neck cancer and delve deeper into patients’ symptoms by asking more relevant questions meaning that patients maybe identified earlier in their disease meeting the Scottish Government’s policy of earlier identification of cancer.
- The pharmacists noted that the pathway fitted in well with their current practice and did not add much additional workload making its easy to incorporate into their service.
- The project has highlighted the additional services that a pharmacy can provide and this may lead to patients accessing pharmacies for advice rather than turning up at A&E if they cannot get to see a GP, which may help to reduce pressure on acute services.
- The project demonstrated that the work of pharmacy teams in this area is broadly welcomed by GP’s and oncologists which will help to increase communication between the different disciplines.
- The Centre for Sustainable Delivery’s Earlier Cancer Diagnosis Programme Board has been supportive of the project and have engaged with the Scottish Government’s Pharmacy Policy Team, to explore the route to national implementation.
Benefits to MSD
- Enhanced reputation of MSD through partnership work. NHS stakeholders completed a survey following the project and the results are shown below:
- Average score of 9.3/10 was scored for being satisfied with MSD’s collaborative working project and 9/10 believed MSD contributed to their organisation’s cancer service pathway respectively.
- NHS stakeholders were asked prior to working with MSD on this collaborative working project, how likely were you to work with MSD or the pharmaceutical industry and the response was 4.3/10. Whereas after the project was completed, the score went to 9/10.
- Quotes taken from the survey when questioned, “what has improved in your service/pathway following this project–
- “MSD’s support enabled us to take forward a project that we would have been unable to deliver otherwise. The project has opened up a discussion about the possibility of a National Service in Scotland.”
- “This was a feasibility project which has hopefully provided some groundwork and evidence for consideration of further roll out of the service.”
- When questioned on feedback of your experience of working in collaboration with MSD –
- “MSD and its officers were helpful and supportive and contributed very positively to the experience of delivering the project. Mr MacDonald was/is a pleasure to work with.”
- “My MSD contact was very supportive and, aside from financial support, helped to facilitate communication with relevant contacts, provide background data and assist in development of the project plan and obtaining of approvals.”
- This collaboration may have enabled more patients to access innovative treatments in line with SMC guidance which may or may not have included MSD medicines.
Funding & Resources
This project was a shared funding commitment from NHS Tayside & MSD. The total project cost was £36,222.
Publications
No publications planned at this time.
GB-NON-09808 | August 2024
Kidney – Active Projects
The Clatterbridge Redesigning Renal Cancer Services
Project Title
The Clatterbridge Redesigning Renal Cancer Services
Organisations involved
The Clatterbridge Cancer Centre NHS Foundation Trust & MSD
Summary
The project aims to increase the Advanced Practitioner (ANP) Whole Time Equivalent (WTE) from 0.6 to 1.0 and increase the number of personal assistants to enable the delivery of a redesigned and more efficient Renal Cell Carcinoma (RCC) service. This increase in clinical capacity will allow the ANP to:
- Optimise the use of follow-up slots from the renal clinics and update follow-up protocols for patients under active surveillance and systemic therapy.
- Extend the current clinic to include post-operative care and separate mixed clinics into metastatic and post-operative clinics.
- Redesign the RCC pathway to improve patient management, reduce follow up and referral delays.
- The purpose of MSD providing funding to cover nursing costs for a defined period of 9 months is to allow the Partner to produce a business case, after the conclusion of this project, to seek to have this funding continued and provided by the NHS on a permanent basis. MSD is therefore providing the Partner with limited funding needed to produce proof-of-concept, such that the NHS could assume responsibility for standing up a full-time Advanced Nurse Practitioner on a permanent basis thereafter.
Deliverables
The additional funding to support the increase in ANP WTE will directly enable the following deliverables:
- Revised follow-up protocols.
- Enhanced post-operative and metastatic clinic pathways.
- Improved clinic slot availability and reduced time from referral to first appointment.
Benefits
Patient Benefit:
- Improved Clinic Slot Availability: The redesign aims to close the gap between available clinic slots and demand, ensuring patients have timely access to appointments
- Enhanced Patient Experience: The proposal focuses on improving the overall patient journey through the pathway, optimising workforce capacity, and refining referral protocols
- Reduced Wait Times: By optimising follow-up slots and protocols, the redesign will reduce the time from referral to the first appointment, providing patients with quicker access to care
Trust Benefit:
- Optimised Clinic Operations: The redesign aims to reduce clinic overbooking and improve the flow in the post-operative pathway, which will enhance the efficiency of clinic operations and resource utilisation
- Enhanced Workforce Capacity: By increasing the Whole Time Equivalent (WTE) for band 8A ANP and the number of Personal Assistants (PA) per week, the trust will benefit from improved workforce capacity and better support for patient management
- Improved Referral Protocols: The proposal includes reviewing and amending referral protocols to ensure they meet the needs of the patients and the trust, which will streamline the referral process and reduce delays
MSD Benefit:
- Partnership Building: Strengthening collaboration with the NHS and enhancing MSD’s reputation as a trusted partner in healthcare innovation.
- Responsible Business Value Delivery: Demonstrating commitment to improving patient care and contributing positively to the healthcare ecosystem.
- This intended collaboration may enable more patients to have access to treatment options in line with NICE guidelines which may or may not include MSD medicines
Funding & Resources
This project is a shared contribution between The Clatterbridge Cancer Centre NHS Foundation Trust and MSD. MSD will fund a total of £36,400 with £22,000 covering the 0.4 Whole Time Equivalent (WTE) Band 8A nurse and the rest toward the project management provided.
GB-NON-12384 | January 2026
NHS Greater Glasgow & Clyde (GG&C) Renal Call Cancer Pathway Development Programme
Project Title
NHS Greater Glasgow & Clyde (GG&C) Renal Call Cancer Pathway Development Programme
Organisations involved
NHS Greater Glasgow & Clyde and MSD
Summary
There is an opportunity in NHS Greater Glasgow & Clyde to optimise the renal cell cancer (RCC) pathway to improve service quality, service efficiency, productivity and patient experience. The desired outcome of this project is to improve the efficiency of, or components of the RCC diagnostic and treatment pathways versus current baseline, including the achievement of the 62- and 31-day cancer waiting time targets that include time to surgery, radiotherapy and systemic anti-cancer therapy. The project is intended to run for approximately 12 months.
Background
The project is required to help address increasing pressure on the RCC pathway, particularly in reducing waiting times for surgery and access to systemic anti-cancer therapy (SACT). Despite improvements in quality of care and patient outcomes over the last ten years, the Renal Cancer Quality Performance Indicators for Scotland report in 2022 still highlights several areas of improvement across the pathway including delays in the pathway due to capacity issues delaying transit of patients through the pathway. Consultants, nurses and pharmacists are currently operating over their capacity and demand on services is expected to increase in the coming years.
Project Objectives
- An understanding/baseline of the existing timescales of each speciality in the RCC pathway through a pathway review across the multi-disciplinary teams.
- An understanding and identification of any inefficiencies in the pathway that are contributing to delayed patient transit through the pathway.
- A map of the existing pathway and a gap analysis which will be taken forward by NHS GG&C to help reduce any identified inefficiencies and potentially improve outcomes for RCC patients.
- An improvement in the efficiency of the pathway, or components of the pathway, versus current baseline including the 62-day and 31-day cancer waiting times targets that include time to surgery, radiotherapy and Systemic Anti-Cancer Therapy (SACT).
Benefits
Benefits/ Impact to patients
- An improved patient experience through the RCC pathway in NHS GG&C
- Quicker time to treatment through improved pathway efficiencies
Benefits to the NHS partner
- Smoother & quicker referral process through the RCC pathway from urgent suspected cancer referral to first treatment, leading to more patients being treated in a faster time
- Improvement of the 62-day and 31-day cancer waiting time targets
Benefits to MSD
- Better understanding of the patient journey through the RCC pathway
- Enhanced reputation of MSD through partnership work
- As a pharmaceutical manufacturer of oncology medicines, an indirect result of an improved pathway may be that MSD see usage of their SMC reimbursed medicine increase.
Funding & Resources
This project is a shared contribution of time between NHS Greater Glasgow & Clyde and MSD.
GB-NON-11986 | October 2025
NHS Lothian Renal Cell Cancer Pathway Development Programme
Project Title
NHS Lothian Renal Cell Cancer Pathway Development Programme
Organisations Involved
NHS Lothian and MSD
Summary
There is an opportunity in NHS Lothian to optimise the renal cell cancer (RCC) pathway to improve service quality, service efficiency, productivity and patient experience. The desired outcome of this project is to improve the efficiency of, or components of the RCC pathway versus current baseline, including the achievement of the 62- and 31-day cancer waiting time targets that include time to surgery, radiotherapy and systemic anti-cancer therapy. The project is intended to run for approximately 12 months.
Background
The project is required to help address increasing pressure on the RCC pathway, particularly in reducing waiting times for surgery and access to systemic anti-cancer therapy (SACT). Despite improvements in quality of care and patient outcomes over the last ten years, the Renal Cancer Quality Performance Indicators for Scotland report in 2022 still highlights a number of areas of improvement across the pathway. Additionally, healthcare professionals, including consultants, nurses, and pharmacists, are currently overextended, with expectations of rising demand for services in the future.
Project Objectives
The project aims to establish a baseline understanding of the current timelines for each segment of the RCC pathway through a comprehensive review involving multi-disciplinary teams. It seeks to identify inefficiencies that contribute to delays in patient transit along the pathway and produce a detailed map of the existing pathway along with a gap analysis. NHS Lothian may use the outputs to address identified inefficiencies, potentially improving outcomes for RCC patients and supporting the achievement of the 62- and 31-day cancer waiting time targets.
Benefits
Benefits to Patients
- An improved patient experience through the RCC pathway in NHS Lothian.
- Potentially quicker time to treatment through improved pathway efficiencies.
Benefits to NHS Lothian
- Smoother and quicker referral process through the RCC pathway from urgent suspected cancer referral to first treatment.
- Potential improvement of the 62-day and 31-day cancer waiting time targets
- Potential efficiencies leading to more patients being treated in a faster time.
Benefits to MSD
- Better understanding of the patient journey through the RCC pathway.
- Enhanced reputation of MSD through partnership work.
- As a pharmaceutical manufacturer of oncology medicines, an indirect result of an improved pathway may be that MSD see usage of their SMC reimbursed medicines.
Funding & Resources
This project is a shared contribution of time between NHS Lothian and MSD. No funding is associated with this collaborative working project.
GB-NON-11825 | August 2025
Greater Manchester, East and Mid Cheshire Trusts Pathway Development Programme
Project Title
Greater Manchester, East and Mid Cheshire Trusts Pathway Development Programme
Organisations Involved
The Christie NHS Foundation Trust and MSD
Summary
The purpose of this project is to optimise the Renal Cell Carcinoma (RCC) Patient Pathway from diagnosis across all Urology sites in Greater Manchester, East and Mid Cheshire to treatment being given in the Oncology department at The Christie. This will be achieved through pathway mapping workshops, a GAP analysis workshop and action planning through the implementation of the project.
Background
There is an opportunity in the Greater Manchester region to optimise the RCC pathway to improve the service quality, service efficiency and patient experience. The desired outcome of this project is an improved RCC pathway, achievement of the renal cell carcinoma 31-day Cancer Waiting Time (CWT) Targets and a clear and standardised approach to referral for post-operative management care.
Why the project is needed:
- Patients aren’t offered optimal treatment in a timely manner
- Lack of a consistent approach to referring RCC patients for treatment
- Standardisation of Specialist Multidisciplinary Team pathway
Project Objectives
The aim of this project is to improve patient experience through promoting quality cancer care and ensuring all renal cell carcinoma patients receive optimal cancer care.
Benefits
Patient Benefits
- An improved patient experience of the renal cancer pathway.
- Earlier treatment of renal cancer patients, improving the chance of successful patient outcome.
Benefits to the NHS Partner
- Achievement of the renal cancer 31-day treatment target CWT.
- Earlier treatment of renal cancer patients, improving patient outcomes.
Benefits to MSD
- Enhanced reputation of MSD through partnership work.
- As a pharmaceutical manufacturer of oncology medicines, an indirect result of an improved pathway may be that MSD see more appropriate usage of their NICE/SMC approved medicines.
Funding & Resources
No funding is associated with this project. This project is a shared contribution of time between The Christie NHS Foundation Trust and MSD.
GB-NON-11423 | June 2025
Kidney – Completed Projects
NHS Tayside Renal Cell Cancer Pathway Development Programme
Project Title
NHS Tayside Renal Cell Cancer Pathway Development Programme
Organisations involved
MSD, NHS Tayside
Summary
There was an opportunity in NHS Tayside to optimise the renal cell cancer (RCC) pathway to improve service quality, service efficiency, productivity and patient experience. The desired outcome of this project was to improve the efficiency of, or components of, the RCC pathway versus the current baseline, including the achievement of the 62- and 31-day cancer waiting time targets that included time to surgery, radiotherapy and systemic anti-cancer therapy. The project was intended to run for approximately 12 months.
Benefits Realised
Due to significant and unforeseen capacity pressures within NHS Tayside that required prioritisation, it has not been possible to complete the project or realise the intended outcomes of the NHS Tayside RCC Pathway Development Programme.
This collaboration may have enabled more patients to access innovative treatments in line with SMC guidance which may or may not have included MSD medicines.
Funding & Resources
This was a project with shared time commitment from NHS Tayside & MSD.
Lessons learnt
Creation of a formal contingency plan should any of the key stakeholders and MSD project manager have time capacity issues or their organisation suffers significant disruption which affects stakeholder capacity during the implementation of the project.
Publications
No publications are planned.
GB-NON-12783 | April 2026
Streamlining Urology MDT meetings across the Southwest Urology Network of Hospitals
Project Title
Streamlining Urology MDT meetings across the Southwest Urology Network of Hospitals
Organisations involved
Peninsula Cancer Alliance and MSD
Summary
MSD worked in partnership with Peninsula Cancer Alliance (PCA) and the Southwest Urology network, to provide project management support to assess the performance of multi-disciplinary team meetings (MDTMs) across the region with a view to running improvement initiatives depending on the findings. After gaining endorsement and advocacy from the NHS Southwest Urology network, we assessed baseline measurements for each of the meetings to define their current state. Based on these findings we then worked with multidisciplinary stakeholders from each of the teams to utilise MDT streamlining methods and principles to optimise their meetings and, where possible, we measured the impact of doing so.
Benefits Realised
Patient Benefits
- Patients now experience faster clinical decision making because of improved processes and more efficient systems as described below
- Approximately 5,991 patient lives1 have been positively impacted through improved MDT meeting processes across the Peninsula Cancer Alliance
- A faster cancer pathway as per the cancer waiting time standard performance described below
- Patients’ outcomes may potentially improve as a result
NHS Benefits
University Hospitals Plymouth
- Meeting time was saved by reviewing Prostate patients in a separate, parallel session to the main Urology MDT, improving efficiency
- Pathologist capacity was optimised by scheduling patients requiring pathology input at the start of the MDT, allowing the Pathologist to leave once their cases were reviewed
- Five root causes of inaccurate referrals were identified and resolved, leading to time savings in MDT meetings and more efficient processes:
- Patients referred who haven’t had a scan
- Patients referred to imaging review where imaging hasn’t been reported
- Referrals stating – ‘review imaging’ without specifics
- Referrals stating – ‘review histology’ without specifics
- Patients reviewed the previous week that have potentially been added to the list as e.g. Admin error.
- For root causes 3 and 4 the solution was to engage junior doctors to ensure specifics are stated on referral
- For root causes 1, 2 and 5 we engaged imaging secretaries to screen referrals against these criteria and email the referring clinician back when required. A guidance document was created disseminated to support this action
- Changes to the haematuria pathway resulted in fewer urograms being conducted, leading to less incidental findings needing to be discussed at MDT, meaning less ‘traffic’ going through MDT
- Team defined new standardised proforma for EPIC roll-out which will contain drop down choices, ensuring accuracy and standardisation of referral request. This will reduce inaccuracies and omissions, saving MDT coordinator time chasing missing data.
- Identified that SoC couldn’t be implemented as the quality of referrals to MDT would need to be improved first (linked to standardised proforma)
Royal Cornwall
- Identification of meeting best practices from the Cornwall urology MDT and creation of a best practice slide deck that has been shared across regional stakeholders detailing the Cornwall approach to continuous improvement around MDT streamlining initiatives
- Dissemination of the Royal Cornwall Standards of Care to protocolise patients – shared as best practice
- Exploration and use-case description of digital MDT triage and automated application of standards of care
Torbay and South Devon Hospital
- Creation of an SOP defining the new triaging process and roles & responsibilities
- Agreement and creation of Torbay specific Standards of Care to protocolise urology patients
- MDT stakeholders now implementing a pre-meeting to triage patients against Torbay specific Standards of Care (measurements of time saved is now beyond the scope of this project due to closing date)
- Fall-back option agreed in event that clinical lead is not available to run the triage meeting
Royal Devon Hospital
- EPIC short cuts implemented to improve the consistency of oncology specific referrals and outcome reporting for nurses
- Alignment with primary care over inappropriate referrals for suspected prostate cancer ultimately releasing capacity from fewer referrals
- Alignment on need for a ‘one-time patient discussion’, and reducing the number of times patients come back to MDT for discussion
- Unification of active surveillance protocols with both sites now implementing a stratified cancer surveillance programme (STRATCANS)
- Expansion of prostate standards of care across both sites
- Measurements of time saved via the above changes is now beyond the scope of this project due to closing date
- The Peninsula Cancer Alliance will continue to monitor the improvement initiatives being implemented and the befits realised
62-day Cancer Waiting Time Performance summary – Urology Jan-May 2025 (vs Jan-May 2024 baseline)
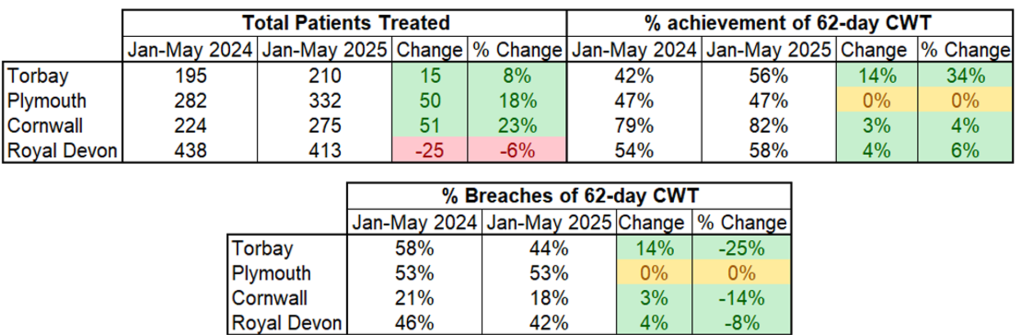
From January to May in 2025 vs baseline:
- Patient volume increased at all Trusts except Royal Devon
- All Trusts improved their performance vs 62-day CWT standard except Plymouth where there was no change
- All Trusts therefore had corresponding decreases in their number of breaches (except Plymouth)
MSD Benefits
- MSD have been able to support continuous quality improvement of urology MDT meetings within the Peninsula Cancer Alliance region, gaining experience and insight into MDT decision making processes
- This collaboration may have enabled more patients to access innovative treatments in line with NICE guidance which may or may not have included MSD medicines
Funding & Resources
- This was a project with shared time commitment from the Peninsula Cancer Alliance & MSD
Lessons learnt
- The project running time was extended by 6 months due to delays in booking a regional clinical urology advisory network meeting where the project was introduced and key engagements and commitments to implementation were gained
- A continuous improvement mindset and approach serves MDT streamlining programmes well, as making changes to the MDT meeting processes requires all stakeholders to be aligned and leadership to come from clinicians who are very limited on time
- The implementation period of the project concluded before data could be captured quantifying efficiencies made. The numerical results are estimates from the clinicians involved and are anecdotal in nature.
Publications
- No intended publications
1 NHSE Cancer Waiting Times Data for 2024 filtered for Urological Cancers and 31 Day Treatment Standard across all 4 Hospitals
GB-NON-11850 | September 2025
South Eastern Health and Social Care Trust Renal Cell Carcinoma Pathway Development Project (PDP)
Project Title
South Eastern Health and Social Care Trust Renal Cell Carcinoma Pathway Development Project (PDP)
Organisations involved
MSD
South Eastern Trust Health and Social Care Trust
Summary
There was an opportunity in South Eastern Health and Social Care Trust to optimise the RCC cancer pathway to improve the service quality, service efficiency, productivity and patient experience. The desired outcome of this project was an improved RCC cancer pathway and achievement of the RCC 31 day and 62 day Cancer Waiting Time (CWT) targets. The project started in March 2023 and finished in February 2024.
Benefits Realised
Through implementation of MSD’s Pathway Development Programme with the South Eastern Health and Social Care Trust team, MSD provided project management support to assess the current state of the RCC cancer pathway and provide a gap analysis of the pathway. MSD then supported the implementation of improvement initiatives to close these gaps and assisted with the measurement of their impact. As a result of the project, the following benefits were realised:
Patient Benefits
- Optimisation of the RCC cancer pathway which allowed patients to navigate through the pathway more efficiently.
- Refer to NHS benefits below to see number of days potentially saved.
NHS Benefits
- Average wait for Computer Tomography (CT), Pathology and Multi – Disciplinary Team (MDT) discussion of results was reduced to 2.5 weeks in 2023 impacting approximately 52 patients
- From September 2023 to March 2024 the following reductions in waiting times were achieved in the RCC adjuvant pathway impacting 16 patients who underwent kidney resection:
- Pathology turn around time post nephrectomy from 6 weeks to 3-4 weeks
- CT scan request post triage from 5 weeks to 1-2 weeks
- Results appointment from surgery date from 6-8 weeks to 5 weeks
- Time to MDT discussion post triage from 6 weeks to 1-2 weeks
- Time from nephrectomy to MDT referral to oncology improved from 17 weeks to 8 weeks within the renal cancer patient pathway.
- % of patients achieving CWT
- 31–day target= improved from 92% in 2022 vs 96% in 2023
- 62-day target= improved from 24% in 2022 vs 25% in 2023
- From the results of the project a regional commissioning paper has been submitted to the Department of Health for a stand-alone Regional Renal MDT to be developed
- Overall treatment rates were not measured as part of this Pathway Development Programme (outside the project scope)
MSD Benefits
- Better understanding of RCC patient needs
- Enhanced reputation of MSD through partnership work. NHS stakeholders completed a survey following the PDP and the results are shown below:
- Average score of 9.5/10 achieved with regards to level of satisfaction of MSD’s collaborative working project.
- Average score of 8.5/10 achieved regarding belief that MSD contributed to improvements in their organisation’s cancer service pathway.
- Average score of 9.5/10 would recommend working in collaboration with MSD to others.
- Quotes taken from the survey when questioned, “what has improved in your service/pathway following this collaborative working project” from NHS Stakeholders:
- “Looking at reducing waiting times for patients such as being referred for CT scan prior to first appointment”
- “Significant improvement in patient pathway resulting in timely referral for consideration of systemic anti-cancer therapy”
- ”Heightened awareness of importance of meeting target timelines”
- When questioned on feedback of your experience of working in collaboration with MSD:
- “Perception of working with a pharmaceutical company would have been sceptical prior to working on this project as felt pressured to use their drugs but there was no pushing of their drugs whatsoever. It was good to introduce an outside source to help get people talking and drill down into the issues. Sometimes the mountain seems too big but we were able to get a few small wins which boosts morale and helps patients in the long run”
- “Extremely professional and patient focused throughout the project”
- This collaboration may have enabled more patients to access innovative treatments in line with NICE guidance which may or may not have included MSD medicines
Funding & Resources
This was a project with shared time commitment from South Eastern Health and Social Care Trust & MSD
Lessons learnt
- Communication is key between all parties which is especially important between departments to optimise decision making and establish the appropriate intervention for the patient as early as possible
- Regular touch points to ensure clarity of expectation and outcomes
- Important to have a clear plan of action and review within stakeholder meetings
- Despite the best efforts of the NHS and MSD to optimise the RCC patient pathway, attainment of the Cancer Waiting Time (CWT) targets remains challenging. The CWT performance 31-day and 62-day for RCC have improved against a backdrop of decreasing performance for other tumour types suggestive of even greater challenges within the system
Publications
There are currently no plans to publish the outcomes of this project by the hospital trust as per the date of this summary.
GB-NON-09804 | November 2024
Western Health and Social Care Trust Renal Cell Carcinoma Pathway Development Project (PDP)
Project Title
Western Health and Social Care Trust Renal Cell Carcinoma Pathway Development Project (PDP)
Organisations involved
MSD
Western Health and Social Care Trust
Summary
There was an opportunity in Western Health and Social Care Trust to optimise the RCC cancer pathway to improve the service quality, service efficiency, productivity and patient experience. The desired outcome of this project was an improved RCC cancer pathway and achievement of the RCC 31 day and 62 day Cancer Waiting Time (CWT) targets. The project started in February 2023 and finished in December 2023.
Benefits Realised
Through implementation of MSD’s Pathway Development Programme with the Western Health and Social Care Trust team, MSD provided project management support to assess the current state of the RCC cancer pathway and provide a gap analysis contrasting the pathway with the national cancer pathway. MSD then supported the implementation of improvement initiatives to close these gaps and assisted with the measurement of their impact. As a result of the project, the following benefits were realised:
Patient Benefits
- Optimisation of the RCC cancer pathway which allows patients to navigate through the pathway more efficiently.
- Refer to NHS benefits below to see number of days potentially saved.
NHS Benefits
- Time from presentation to Computed Tomography (CT) kidney triple phase scan improved from a median of 9 days 2022 to a median of 7.5 days 2023.
- Pathology post-surgery turnaround times maintained at a median of 16 days in both 2022 and 2023.
- New post-surgery RCC pathway implemented across the region allowing all eligible patients to be discussed at the regional specialist urology MDM (Multi-Disciplinary Meeting).
- This project supports the long-term aim of a regional renal service focused around a single regional renal MDT and a regional waiting list based on this.
- Theatre capacity – maintained at 84 sessions in 2022 and 85 sessions in 2023.
- % of patients achieving CWT:
- 31–day = improved from 69% in 2022 vs 73% in 2023
- 62-day = 80% in 2022 vs 61% in 2023
- Overall treatment rates were not measured as part of this Pathway Development Programme
MSD Benefits
- Better understanding of RCC patient needs
- Enhanced reputation of MSD through partnership work. NHS stakeholders completed a survey following the PDP and the results are shown below:
- Average score of 8/10 and 7/10 was scored for being satisfied with MSD’s collaborative working project and believed MSD contributed to improvements in their organisation’s cancer service pathway respectively.
- Average score of 8/10 would recommend working in collaboration with MSD to others.
- Quotes taken from the survey when questioned, “what has improved in your service/pathway following this collaborative working project” from NHS Stakeholders:
- “Standardisation of renal cancer care throughout the region”
- “The pathway already worked very well, however the project was good to bring the team together and highlight the need for regional MDM”
- When questioned on feedback of your experience of working in collaboration with MSD
- “Worked well”
- “Good collaborative working and well organised”
- This collaboration may have enabled more patients to access innovative treatments in line with NICE guidance which may or may not have included MSD medicines.
Funding & Resources
This was a project with shared time commitment from Western Health and Social Care Trust & MSD
Lessons learnt
- Communication is key between all parties which is especially important between departments to optimise decision making and establish the appropriate intervention for the patient as early as possible
- Regular touch points to ensure clarity of expectation and outcomes
- Clear plan of action and review within stakeholder meetings
- Despite the best efforts of the NHS and MSD to optimise the RCC patient pathway, attainment of the Cancer Waiting Time (CWT) targets remains challenging. The CWT performance for RCC however has improved for 31-day target against a backdrop of decreasing performance for other tumour types suggestive of even greater challenges within the system.
Publications
There are currently no plans to publish the outcomes of this project by the hospital trust as per the date of this summary.
GB-NON-09637 | September 2024
Belfast Health and Social Care Trust Renal Cell Carcinoma Pathway Development Project (PDP)
Project Title
Belfast Health and Social Care Trust Renal Cell Carcinoma Pathway Development Project (PDP)
Organisations involved
MSD
Belfast Health and Social Care Trust
Summary
There was an opportunity in Belfast Health and Social Care Trust to optimise the RCC pathway to improve the service quality, service efficiency, productivity and patient experience. The desired outcome of this project was an improved RCC pathway and achievement of the RCC 31-day and 62-day Cancer Waiting Time (CWT) targets. The project started in July 2022 and finished in December 2023.
Benefits Realised
Through implementation of MSD’s Pathway Development Programme with the Belfast Health and Social Care Trust team, MSD provided project management support to assess the current state of the RCC pathway and provide a gap analysis contrasting the pathway with the national cancer pathway. MSD then supported the implementation of improvement initiatives to close these gaps and assisted with the measurement of their impact. As a result of the project, the following benefits were realised:
Patient Benefits
- Optimisation of the RCC cancer pathway which allows patients to navigate through the pathway more efficiently.
- Refer to NHS benefits below to see number of days potentially saved.
NHS Benefits
- Time from Renal Surgery to Multi-Disciplinary Meeting (MDM) improved from 42 days in 2022 to 34 days in 2023.
- Development of a new regional adjuvant RCC pathway developed to meet the 12 week (84 days) patient criteria from nephrectomy to oncology treatment impacting 71 patients from October 2022 to December 2023. The following mean times were achieved:
- Nephrectomy to MDM discussion was 35 days
- Urology results post-surgery appointment 44 days
- Oncology appointment 60 days
- Nephrectomy to Computed Tomography (CT) scan 44 days
- Nephrectomy to start of oncology treatment 82 days
- This project supports the long-term aim of a regional renal service focused around a single regional renal Multi-Disciplinary Team (MDT)
- Improvement in cancer Waiting Times
- 31–day = improved from 40% in 2022 vs 51% in 2023
- 62-day = 6% in 2022 vs 7% in 2023
- Overall treatment rates were not measured as part of this Pathway Development Programme
MSD Benefits
- Better understanding of RCC cancer patient needs
- Enhanced reputation of MSD through partnership work. NHS stakeholders completed a survey following the PDP and the results are shown below:
- Average score of 9/10 and 8/10 was scored for being satisfied with MSD’s collaborative working project and believed MSD contributed to improvements in their organisation’s cancer service pathway respectively.
- Average score of 7/10 would recommend working in collaboration with MSD to others.
- Quotes taken from the survey when questioned, “what has improved in your service/pathway following this collaborative working project” from NHS Stakeholders:
- “Regional renal cancer multi-disciplinary team being planned. Supported by clinical staff and regional cancer network”
- “Two of the 14 project measures have progressed through the renal optimisation project”
- “Automated mechanism to flag eligible patients for adjuvant treatment and waiting times met”
- “All other actions will remain on the trusts cancer improvement plan for regular review”
- “Improved discussion regarding the issues and potential solutions”
- When questioned on feedback of your experience of working in collaboration with MSD
- “Excellent, provided drive and support to a project that would not otherwise have happened”
- “This project has allowed us to produce a plan for the improvements required across the renal pathway”
- “Helpful to have outside input to drive things forward”
- This collaboration may have enabled more patients to access innovative treatments in line with NICE guidance which may or may not have included MSD medicines
Funding & Resources
This was a project with shared time commitment from Belfast Health and Social Care Trust & MSD
Lessons learnt
- Communication is key between all parties which is especially important between departments to optimise decision making and establish the appropriate intervention for the patient as early as possible
- Regular touch points to ensure clarity of expectation and outcomes
- Clear plan of action and review within stakeholder meetings
- Despite the best efforts of the NHS and MSD to optimise the RCC patient pathway, attainment of the Cancer Waiting Time (CWT) targets remains challenging. The 31 day CWT performance for RCC has improved against a backdrop of decreasing performance for other tumour types suggestive of even greater challenges within the system. It is unlikely the actions taken as part of the project contributed to the increase in 31 day performance as the main actions implemented related to improving the pathway between first and subsequent treatment
Publications
There are currently no plans to publish the outcomes of this project by the hospital trust as per the date of this summary.
GB-NON-09635 | September 2024
Southern Health and Social Care Trust Renal Cell Carcinoma Pathway Development Project (PDP)
Project Title
Southern Health and Social Care Trust Renal Cell Carcinoma Pathway Development Project (PDP)
Organisations involved
MSD
Southern Health and Social Care Trust
Summary
A national optimal Renal Cell Carcinoma (RCC) cancer pathway has been created with the intention of improving patient experience through promoting quality cancer care and ensuring all RCC patients receive optimal cancer care. There was an opportunity in Southern Health and Social Care Trust to optimise the RCC cancer pathway to improve the service quality, service efficiency, productivity and patient experience. The desired outcome of this project was an improved RCC cancer pathway and achievement of the RCC 31 day and 62 day Cancer Waiting Time (CWT) targets. The project started in July 2022 and finished in December 2023.
Benefits Realised
Through implementation of MSD’s Pathway Development Programme with the Southern Health and Social Care Trust team, MSD provided project management support to assess the current state of the RCC cancer pathway and provide a gap analysis of the pathway. MSD then supported the implementation of improvement initiatives to close these gaps and assisted with the measurement of their impact. As a result of the project, the following benefits were realised:
Patient Benefits
- Optimisation of the RCC cancer pathway which allows patients to navigate through the pathway more efficiently. This could potentially increase the chance of a successful outcome if patients are diagnosed earlier on in the pathway.
- Refer to NHS benefits below to see number of days potentially saved.
NHS Benefits
- All RCC patients in 2022/2023 now triaged within 0-3 days impacting 60 patients
- Time to surgery improved from 41 days in April 2023 to 21 days in December 2023 impacting 36 patients
- Time to post surgery Computed Tomography (CT) scan improved from 23 days in April 2023 to 13 days in December 2023
- Time to first surgical outpatients appointment post-surgery improved from 26 days in March 2023 to 19 days in December 2023
- This project supports the long-term aim to develop a regional renal service focused around a single regional renal MDT.
- Two nurses can now directly request CT Urograms following protocol training (the time aspect of this has not been measured)
- % of patients achieving CWT
- 31–day target= improved from 82% 2022 vs 86% 2023
- 62-day target= declined from 17% 2022 vs 0% 2023
- Overall treatment rates were not measured as part of this Pathway Development Programme (outside the project scope)
MSD Benefits
- Better understanding of RCC patient needs
- Enhanced reputation of MSD through partnership work. NHS stakeholders completed a survey following the PDP and the results are shown below:
- Average score of 6.75/10 achieved with regards to level of satisfaction of MSD’s collaborative working project.
- Average score of 5.25/10 achieved regarding belief that MSD contributed to improvements in their organisation’s cancer service pathway.
- Average score of 5.75/10 would recommend working in collaboration with MSD to others.
- Quotes taken from NHS stakeholders when questioned, “what has improved in your service/pathway following this collaborative working project”:
- “Streamlined pathway for patients post-surgery. More collaborative working across teams and increased emphasis on getting patients through the pathway”
- “Multi-disciplinary work on addressing priority areas following the process mapping work. Engagement of staff from different areas / specialities to see feedback”
- Quotes taken from NHS stakeholders when questioned on “feedback of your experience of working in collaboration with MSD”:
- “Good partnership working. Facilitation of meetings were very good and it was useful to have an independent person involved in this”
- “Clear objectives, excellent communication, regular updates. Encouraged more multi team working. Professional”
- This collaboration may have enabled more patients to access innovative treatments in line with NICE guidance which may or may not have included MSD medicines
Funding & Resources
This was a project with shared time commitment from Southern Health and Social Care Trust & MSD
Lessons learnt
- Communication is key between all parties which is especially important between departments to optimise decision making and establish the appropriate intervention for the patient as early as possible
- Regular touch points to ensure clarity of expectation and outcomes
- Important to have a clear plan of action and review within stakeholder meetings
- Despite the best efforts of the NHS and MSD to optimise the RCC patient pathway, attainment of the Cancer Waiting Time (CWT) targets remains challenging especially the 62 day CWT due to system capacity pressures. The CWT performance 31-day for RCC has improved against a backdrop of decreasing performance for other tumour types suggestive of even greater challenges within the system
Publications
There are currently no plans to publish the outcomes of this project by the hospital trust as per the date of this summary.
GB-NON-09636 | July 2024
Lung – Active Projects
West Midlands Cancer Alliance (WMCA) Lung Cancer Pathway Development Programme (PDP)
Project Title
West Midlands Cancer Alliance (WMCA) Lung Cancer Pathway Development Programme (PDP)
Organisations involved
West Midlands Cancer Alliance (WMCA)
George Eliot Hospital NHS Trust
Walsall Healthcare NHS Trust
Worcestershire Acute Hospitals NHS Trust
Shrewsbury & Telford Hospital NHS Trust
MSD (Merck Sharp & Dohme (UK) Limited)
Summary
The intent of this collaboration is to optimise the lung cancer pathway through pathway mapping and gap analysis workshops. This will be delivered across four NHS Trusts within the WMCA footprint: George Eliot Hospital NHS Trust, Walsall Healthcare NHS Trust, Worcestershire Acute Hospitals NHS Trust, Shrewsbury & Telford Hospital NHS Trust. The collaboration will focus on improving Cancer Waiting Times (CWT) including the 28-day, 31-day & 62-day standards, through optimising the quality, efficiency, and productivity of the pathway. This will support lung cancer patients to access timely services and treatments, improving patient experience and outcomes.
This project will be delivered collaboratively by WMCA, participating NHS trusts, and MSD through the co-development and delivery of pathway mapping and gap analysis workshops. These workshops will be used to identify challenges and barriers, improve understanding of local needs, and inform potential solutions to optimise the lung cancer pathway.
The project intends to run for a duration of 12 months. Based on the findings from the gap analysis, both partners will subsequently work with the NHS trusts towards implementing plans addressing the identified areas of improvement and commit to measuring the outcomes of the project and disseminating these outcomes within 6 months of the project completion.
Background
Following an internal review by WMCA Lung Expert Advisory Group (LEAG), there is an opportunity across WMCA to optimise the lung cancer pathway to improve outcomes for lung cancer patients in the West Midlands. There are concerns due to pathway gaps, blockages, inefficiencies & variations, that not all patients are being diagnosed or referred to the Multi-Disciplinary Team (MDT) and Oncology clinical teams in a timely manner. This may result in patients missing out on timely access to treatment, which will negatively impact patient outcomes and experience.
This opportunity will aim to ultimately support the achievement of the NHS CWT 28-day, 31-day & 62-day treatment targets, as well as a reduction in time taken and increased number of patients receiving SACT treatment. This collaboration has the potential to make improvements to patient experience and outcomes in line with the national cancer plan.1
Project Objectives
- Optimisation of lung cancer patient pathway across four trusts within the WMCA footprint, reducing variation in cancer care and treatments
- Understand the challenges, bottlenecks and inefficiencies within the lung cancer patient pathway
- Identify ways to address the challenges and obstacles within the patient pathway to optimise the pathway
- Identify and reduce inconsistencies seen in the existing patient pathway, reducing variation/health inequalities in care
Benefits
Benefits to patients
- Improved access to services and treatments
- Improved patient outcomes and experience
Benefits to the NHS
- Increased achievement of 28-day, 31-day & 62-day Cancer Waiting Times (CWT) at trust level
- Increase in treatment rates for lung cancer patients
- Addressing health inequalities across the West Midlands region
- WMCA improving SACT rates as a region
Benefits to MSD
- Better understanding of the patient journey through the lung cancer pathway
- Enhanced reputation of MSD through partnership work
- As a pharmaceutical manufacturer of oncology medicines, an indirect result of an improved pathway may be that MSD see more usage of their NICE approved medicines
Funding & Resources
This project is a shared contribution of time between WMCA and MSD.
References
1 Government UK (Gov.UK). The National Cancer Plan for England: delivery world class cancer care. The National Cancer Plan for England: delivering world class cancer care (Accessed April 2026)
GB-NON-12856 | April 2026
Health Equity Nurse (HEN) – Overcoming Health Inequalities in the Lung Cancer Pathway at Barts Health NHS Trust
Project Title
Health Equity Nurse (HEN) – Overcoming Health Inequalities in the Lung Cancer Pathway at Barts Health NHS Trust
Organisations involved
Barts Health NHS Trust
North East London Cancer Alliance (NELCA), c/o North East London Integrated Care Board (ICB)
MSD (Merck Sharp & Dohme (UK) Limited)
Summary
This collaborative working project aims to improve equity of access to active lung cancer treatment, including clinical trial awareness and participation, for diverse and deprived communities served by Barts Health NHS Trust.
The project will establish a dedicated NHS Band 7 Health Equity Nurse (HEN) role for a 15‑month period to provide personalised patient navigation and support across the lung cancer pathway, with a particular focus on patients experiencing health inequalities.
Through targeted patient engagement, education, and pathway navigation support, the project seeks to reduce unwarranted variation in access to care, improve patient experience, and generate robust evidence to inform a future NHS business case for sustainable funding of the HEN role.
Background
Barts Health NHS Trust serves one of the largest, most diverse and socio‑economically deprived populations in the UK. Evidence demonstrates that patients from minority, diverse and deprived communities experience poorer cancer care outcomes, including delays in diagnosis, reduced access to information, and lower uptake of active treatment and clinical trial participation.1
Local pathway data and national evidence highlight the need for tailored non‑clinical interventions to address practical, cultural and socio‑economic barriers to care. The Health Equity Nurse role has been designed to respond to these unmet needs by supporting patients through key pathway stages and improving understanding of barriers to equitable access.
Project Objectives
- More equitable and proportional access for diverse and deprived communities to active lung cancer treatment including clinical trial participation.
- Improved patient experience measured through patient surveys to understand impact of HEN role.
- Demonstration of value and impact of HEN role for diverse and deprived communities.
Benefits
Benefits to patients
- Enhanced support to help patients navigate the lung cancer pathway from first outpatient appointment (OPA) through to treatment planning milestones, supporting timely progression in line with cancer waiting times (CWT) standards and improving equity of access.
- Improved awareness, education, understanding and confidence of clinical trials and navigation of the lung cancer pathway for identified diverse and deprived communities.
Benefits to the NHS
- Less variation in timed pathway from 1st OPA to decision to treat part of the lung cancer pathway for identified diverse and deprived communities.
- Increased capacity for clinical roles (CNS in particular) through additional support provided by Health Equity Nurse role.
Benefits to MSD
- Enhanced reputation of MSD through partnership work.
- As a pharmaceutical manufacturer of oncology medicines, an indirect result of an improved pathway may be that MSD see more appropriate usage of their NICE approved medicines.
Funding & Resources
This project is a shared contribution between Barts Health NHS Trust, North East London Cancer Alliance (NELCA) c/o North East London Integrated Care Board (NEL ICB) and MSD.
The total project cost is £95,707.
References
1 Cancer Research UK (CRUK). Cancer in the UK 2025: Socioeconomic Deprivation. Cancer in the UK 2025 – socio economic deprivation (Accessed February 2026).
GB-NON-12648 | March 2026
Oxford lung pathway development through digital health adoption
Project Title
Oxford lung pathway development through digital health adoption
Organisations involved
Oxford University Hospitals NHS Foundation Trust (OUH) and MSD UK
Summary
This collaborative working project between the OUH and MSD UK aims to improve the Oxford lung cancer pathway through the adoption of two digital health solutions: Axana’s NextGen MDT Platform and the NHS Federated Data Platform’s Cancer 360.
The project will support the Respiratory Early Diagnosis Service (REDS), lung cancer pathway stakeholders and the wider lung cancer multidisciplinary team (MDT) to streamline patient tracking, MDT preparation and decision‑making, and post‑MDT follow‑up. By embedding these digital tools in routine practice, the project intends to shorten the time from patient referral to treatment, reduce delays and breaches within the 62‑day cancer waiting time (CWT) standard, and release clinical and administrative capacity.
Over an initial 12‑month implementation period followed by evaluation, the project will:
- Implement two digital health solutions (from Axana and Cancer 360) within the lung pathway.
- Improve clinician workflow and reduce administrative burden in MDT coordination.
- Shorten the time from patient referral to treatment initiation.
- Evaluate the impact of digital tools on:
- Clinician time savings
- Patient journey duration
- MDT process efficiency
Background
- Despite meeting the 28‑day Faster Diagnosis Standard, OUH currently faces significant delays in the lung cancer pathway after diagnosis. In April 2025, 84% of lung cancer patients breached the 62‑day cancer waiting time standard, with only 31% of patients meeting the national 85% target.1 Bottlenecks in diagnostic and staging scans, specialist scheduling and pathology turnaround times contribute to these delays, with associated risks of disease progression, increased patient anxiety and potential external scrutiny of pathway performance.
- At the same time, demand on cancer services is increasing due to improved detection, early diagnosis programmes and patients living longer with cancer. This drives higher patient volumes and adds pressure to existing capacity. MDT meetings are a particular pressure point; studies have shown that, in many MDTs, around half of patients are discussed for two minutes or less,2 limiting the depth of discussion for complex cases.
- The NHS is moving from analogue to digital ways of working, creating an opportunity for OUH to improve operational efficiency in the lung cancer pathway. Cancer 360 offers an integrated view of patient data across multiple sources, while Axana’s NextGen MDT Platform provides an MDT‑focused workflow solution with AI‑enabled data collation and task management. Together these tools provide a foundation to streamline MDT coordination, reduce administrative burden and improve timely, data‑informed decision‑making for lung cancer patients at OUH.
Project Objectives
Overall aim
By November 2026, we aim to increase the proportion of lung cancer patients at OUH meeting the 62‑day cancer waiting time standard from 31% to 50% by embedding Axana and Cancer 360 within the lung cancer pathway and evaluating their impact. Our key objectives are to:
- Implement digital solutions within the lung pathway
- Implement Axana’s NextGen MDT Platform and Cancer 360 within the OUH lung cancer pathway, including mapping “as‑is” processes and designing the “to‑be” workflows that incorporate these tools.
- Improve clinician workflow and reduce administrative burden
- Reduce time spent by MDT clinicians and coordinators on manual data collection and preparation.
- Improve access to real‑time, centralised patient information to support MDT preparation and in‑meeting decision‑making.
- Shorten time from referral to treatment
- Reduce delays and breaches within the 62‑day lung cancer pathway and improve performance against the 28‑day Faster Diagnosis Standard and 31‑day treatment standards.
- Evaluate the impact of digital tools
- Measure changes in:
- Achievement of 62‑, 31‑ and 28‑day CWT standards,
- Number of breaches, backlog and backstop patients,
- Administration and MDT preparation time,
- Patient throughput and time from decision to treat to outpatient appointment, and
- Patient‑reported experience (PREMs), including equity‑focused metrics aligned to Core20PLUS5.
- Measure changes in:
Benefits
Benefits to patients
- Faster diagnosis and treatment
- Improved care coordination
Benefits to the OUH
- Operational efficiencies within the 62-day pathway and the MDT
- Reduction in pathway delays and breaches
- Capacity released through improved resource utilisation and reduction in administration
Benefits to MSD
- Create a case study for how MSD can support NHS provider Trusts to move from analogue to digital, and adopt and embed new digital health solutions
- Strengthened NHS collaboration with Oxford and Thames Valley Cancer Alliance
- This collaboration may ultimately enable more patients to access innovative treatments in line with NICE guidance which may or may not include MSD medicines.
Funding & Resources
This project is a shared contribution of time between Oxford University Hospitals NHS Foundation Trust and MSD.
1 NHSE Cancer Waiting Times Data – Oxford performance versus the 62-day cancer waiting time standard for lung pathway using April 2025 data
2 Cancer Research UK, “Meeting Patients’ Needs, improving the effectiveness of multidisciplinary team meetings”, January 2017.
GB-NON-12573 | February 2026
Oxford Respiratory Early Diagnosis Service (REDS) Lung Dataset & Dashboard Development
Project Title
Oxford Respiratory Early Diagnosis Service (REDS) Lung Dataset & Dashboard Development
Organisations involved
Oxford University Hospitals NHS Foundation Trust and MSD
Summary
The REDS team currently maintain a database to track people under their care through the lung cancer diagnostic pathway, but the existing dataset is difficult to manage and does not easily generate insights or reports. This collaborative working project between Oxford University Hospitals NHS Foundation Trust and MSD aims to improve the functionality and usability of the REDS dataset and to develop an automated dashboard that provides timely, actionable insights to inform lung cancer service development.
Key elements of the project include:
- Refining the current REDS dataset so it is more user-friendly and supports improved reporting and insight generation.
- Building a REDS dashboard based on the improved dataset, including automation of dashboard updates.
- Training the REDS team, delivered by MSD, to strengthen data management and synthesis skills so they can interrogate their own service data, identify areas for improvement and uncover health inequalities.
The project will run with MSD support for an initial period of approximately six months, using a series of “test and learn” development cycles. It is expected to support service optimisation, enable more efficient reporting to the Trust and Cancer Alliance, and ultimately help patients move through the lung cancer pathway more quickly so they can access appropriate treatments in a timelier manner.
Background
The REDS team are responsible for managing patients as they move through the lung cancer diagnostic pathway. At present, relevant patient information is spread across multiple systems, including Cerner (EPR), Infoflex and Orbit, and the team maintain their own separate REDS database to ensure they have a complete view of patients under their care.
However, the REDS dataset is not currently configured in a way that allows for easy interrogation or report generation. The team lack the tools and capability to synthesise their data efficiently, which means:
- They cannot quickly generate the insights needed to inform pathway development and optimisation.
- There are delays between identifying areas for improvement, implementing pathway changes and seeing tangible impact.
In addition, the REDS team have ongoing reporting requirements to both the Trust and the Thames Valley Cancer Alliance, but the current dataset does not support automated report generation. There is also a need for better visibility of patients across six different referral routes, to ensure that no patients are missed and that investigations and appointments are delivered within appropriate timeframes.
Collectively, these challenges create a gap in the team’s ability to use their own data for operational decision-making, to track health inequalities, and to support achievement of cancer waiting time targets (28-day and 62-day standards).
Project Objectives
For Patients
- Support shorter and more efficient lung cancer diagnostic pathways
- Improve visibility of underserved or under‑represented patient groups
For Oxford University Hospitals NHS Foundation Trust
- Refine and improve the REDS dataset
- Strengthen the REDS team’s capability to interrogate their data, identify bottlenecks and drive focused improvement work, including tackling health inequalities.
- Support delivery of national cancer waiting time standards
For MSD
- Research, build, test and handover an improved REDS dashboard
- Assess scalability and transferability of the approach across other Trusts.
Benefits
Benefits to Patients
- A more optimised and timely diagnostic pathway, with reduced delays and faster access to treatment.
- Improved visibility of patient flow across referral routes, reducing the risk of patients being missed.
- Enable targeted improvements for underserved communities.
Benefits to Oxford University Hospitals NHS Foundation Trust
- Enhanced ability for the REDS team to interrogate their data, generate insights and optimise the pathway more rapidly.
- Automated dashboards and reporting reduce manual workload and support Trust and Cancer Alliance reporting.
- Improved identification of health inequalities to inform targeted interventions.
- A structured benefits realisation framework aligned to project metrics, such as insights generated, operational improvements and time/capacity savings.
Benefits to MSD
- Greater understanding of digital pathway monitoring within NHS services.
- Improved capability in designing and evaluating collaborative optimisation projects.
- Strengthened reputation as a collaborative partner through non-promotional service development work.
- Opportunity to evaluate potential scalability to other Trusts and support more patients reaching appropriate treatment in a timely manner.
Funding & Resources
This project is a shared contribution of time between Oxford University Hospitals NHS Foundation Trust and MSD.
GB-NON-12571 | February 2026
Western Health and Social Care Trust Lung Cancer Pathway Development Programme (PDP)
Project Title
Western Health and Social Care Trust Lung Cancer Pathway Development Programme (PDP)
Organisations involved
MSD
Western Health and Social Care Trust
Summary
There is an opportunity in Western Health and Social Care Trust to optimise the lung cancer pathway to improve the service quality, service efficiency, productivity and patient experience. The desired outcome of this project is potential improvement of the lung cancer patient pathway and of the 62-day* and 31-day** cancer waiting time (CWT) targets, currently set at a target of 95% for 62-day and 98% for 31- day cancer waiting times. The project intends to run for approximately 12 months.
Background
Current Problems & Needs:
- Pressure in the pathway to achieve the 62- and 31-day cancer waiting times, with over 50% of lung cancer patients in Northern Ireland waiting longer than the 62-day target (reference: NI Department for Health Cancer Waiting Times).
- Despite improvements in quality of care and patient outcomes over the last ten years, 44.4% of Northern Ireland lung cancer patients are diagnosed with advance stage IV disease (reference: Northern Ireland Cancer Registry).
- Delays in the pathway due to capacity issues delaying transit of patients through the pathway.
- Consultants, nurses and pharmacists are currently over capacity and demand on services is expected to increase further due to the introduction of lung cancer screening.
Project Objectives
The primary objective of this project is the optimisation of the lung cancer pathway across Western Health and Social Care Trust. Specifically contributing towards; –
- Potentially improving the 62-day and 31-day treatment target cancer waiting time targets
- Measurement of the outcomes of the project and disseminating these outcomes within 6 months of the project completion
Benefits
Patient Benefits
- An improved patient experience through the lung cancer pathway in the Western Health and Social Care Trust
- Potentially quicker time to treatment through improved pathway efficiencies
NHS Benefits
Optimised lung cancer pathway across Western Health and Social Care trust hospital sites resulting in
- More efficient & quicker referral process through the lung cancer pathway from red flag referral to treatment
- Potential improvement of the 62-day and 31-day cancer waiting time targets
- Potential efficiencies leading to more patients being treated in a faster time
MSD Benefits
- Better understanding of the patient journey through the lung cancer pathway
- Enhanced reputation of MSD through partnership work
- As a pharmaceutical manufacturer of oncology medicines, an indirect result of an improved pathway may be that MSD see more appropriate usage of their NICE/SMC reimbursed medicines
Funding & Resources
No funding is associated with this project. This project is a shared contribution of time between Western Health and Social Care Trust and MSD.
* 62-day CWT: 62-day target between the date hospital receives urgent referral for suspected cancer and start of treatment
** 31-day CWT: 31-day target between the date doctor agrees treatment plan and start of treatment
GB-NON-10354 | January 2025
Lung Cancer Healthbot – Greater Manchester Cancer Alliance Self-Referral to Chest X-Ray Symptom Awareness & Activation Project
Project Title
Lung Cancer Healthbot – Greater Manchester Cancer Alliance Self-Referral to Chest X-Ray Symptom Awareness & Activation Project
Organisations involved
- Greater Manchester Cancer Alliance (GM Cancer)
- The Christie NHS Foundation Trust
- MSD UK Ltd
Summary
In 2022 Greater Manchester introduced a Self-Referral Chest X-Ray (SRCXR) service across some of its localities to enable patients who may have worrying respiratory symptoms to have direct access to a chest x-ray. The service has been running well and GM Cancer wishes to ensure more coverage and awareness of the service and thus increase uptake. A campaign in certain communities both out of home and social media could help bring more awareness and coupled with a sustainable method of activating patients to seek help, will aid more patients to attend the SRCXR service. This project will bring together a digital healthbot co-designed with GM Cancer Alliance and the Lung Pathway Board and developed by MSD with an awareness campaign to help more people become aware of potential symptoms of lung cancer and then take help-seeking action to attend the SRCXR service. Attendance rates to the service, reach of and engagement with the awareness campaigns and healthbot will be measured to help understand the impact of the project on the service.
Background
Since July 2022, members of the public registered at a GP in Bury, Heywood, Middleton & Rochdale (HMR) have been able to access a chest x-ray at their local hospital, without having to see a GP, or book an appointment in advance. The scheme not only intends to offer peace of mind for many, who may be experiencing concerns about their health, but to also expose the cases which may need extra attention quickly. Conditions like lung cancer have better outcomes when they are caught earlier[1] , so the pilot intended to improve early diagnosis, and potentially improve outcomes for residents. The initiative is available at select hospitals across the area. GM Cancer acknowledge that the self-referral chest x-ray (SRCXR) service is an effective tool to reach a greater number of patients and thus impact earlier diagnosis rates, positively contributing to the NHS long term plan’s 75% target.
Project Objectives
The primary aim of the project is to provide residents of Bury and HMR localities with a digital tool that will signpost them to local services. The secondary aim of the project is to increase awareness and uptake of the SRCXR service via the co-developed Healthbot and measure its impact. The project will run over 9 months and then be evaluated.
Benefits
Potential Patient Benefits
- Awareness of symptoms relating to lung cancer and awareness of services available
- Patients can validate symptom concerns
Potential NHS Benefits
- Wider awareness of SRCXR service
- Use of a sustainable method to improve patient activation to help seeking behaviour as opposed to telephone calls
- Support the utilisation of SRCXR service
- Potential for increase in disease diagnosis and treatment rates
Potential MSD Benefits
- Testing of the Healthbot in the UK
- Understand the wider use of the tool
- The intended benefits of raising awareness of lung cancer symptoms and encouraging a self-referral to chest x-ray may mean that more patients have access to treatment options in line with NICE/SMC guidance, which may or may not include MSD medicines
Funding
This project is a shared contribution between GM Cancer and MSD. The total project cost is £69,600
GB-NON-08732 | January 2024
References
[1] Cancer Research UK; Survival for Lung Cancer document; last accessed 18th January 2024 Survival for lung cancer | Cancer Research UK
Lung – Completed Projects
Kent & Medway Cancer Alliance Lung Pathway Development Programme (PDP)
Project Title
Kent & Medway Cancer Alliance Lung Pathway Development Programme (PDP)
Organisations involved
- Kent & Medway Cancer Alliance & MSD
Summary
A National Optimal Lung Cancer Pathway (NOLCP) has been created with the intention of improving patient experience through promoting quality cancer care and ensuring all lung cancer patients receive optimal care. There was an opportunity in Kent and Medway Cancer Alliance to optimise the lung cancer pathway in line with the NOLCP to improve the service quality, service efficiency, productivity and patient experience. The desired outcome of this project was an improved lung cancer pathway aligned with the NOLCP and achievement of the lung cancer 28 day Faster Diagnostic Standard (28 Day FDS) and the 31 and 62 day Cancer Waiting Time Targets (CWTs). The aim was to reduce variation and optimise day -3 to day 49 in the delivery of the NOLCP with a specific focus on reducing the variation in care, optimising outcomes and delivering timely pathway performance to achieve diagnosis and treatment to the patient in 4 trusts (5 MDTs) in Kent & Medway Cancer Alliance:
- East Kent Hospitals University NHS Foundation Trust (EKHUFT) (2 MDTs)
- Maidstone & Tunbridge Wells NHS Trust (MTW)
- Medway NHS Foundation Trust (MFT)
- Dartford & Gravesham NHS Trust (DGT)
The Project began on 1st July 2024 and finished on 30th June 2025.
Benefits Realised
Through implementation of MSD’s Pathway Development Programme with Kent & Medway Cancer Alliance, MSD provided project management support to map the current NOLCP, facilitated gap analysis workshops and worked with the Cancer Alliance and each of the 4 Trusts to develop actionable implementation plans to deliver the changes required to achieve the desired outputs. As a result of the project, the following benefits were realised:
Patient Benefits
- Changes in the NOLCP Milestones can be seen in Table 1, with all trusts making a positive impact to reduce the number of days from referral to various NOLCP milestones. Highlights include: The number of days from Referral to First Treatment was reduced across all 4 Trusts. DGT saving 35 days; EKHUFT 12 days; MFT 19.5 days & MTW 7.5 days, a combined total of 74 days saved.
Table 1: Median Waiting Time in Days from Referral to NOLCP Milestone in Lung Pathway: Baseline January – June 2024 vs. Evaluation May – Oct 2025
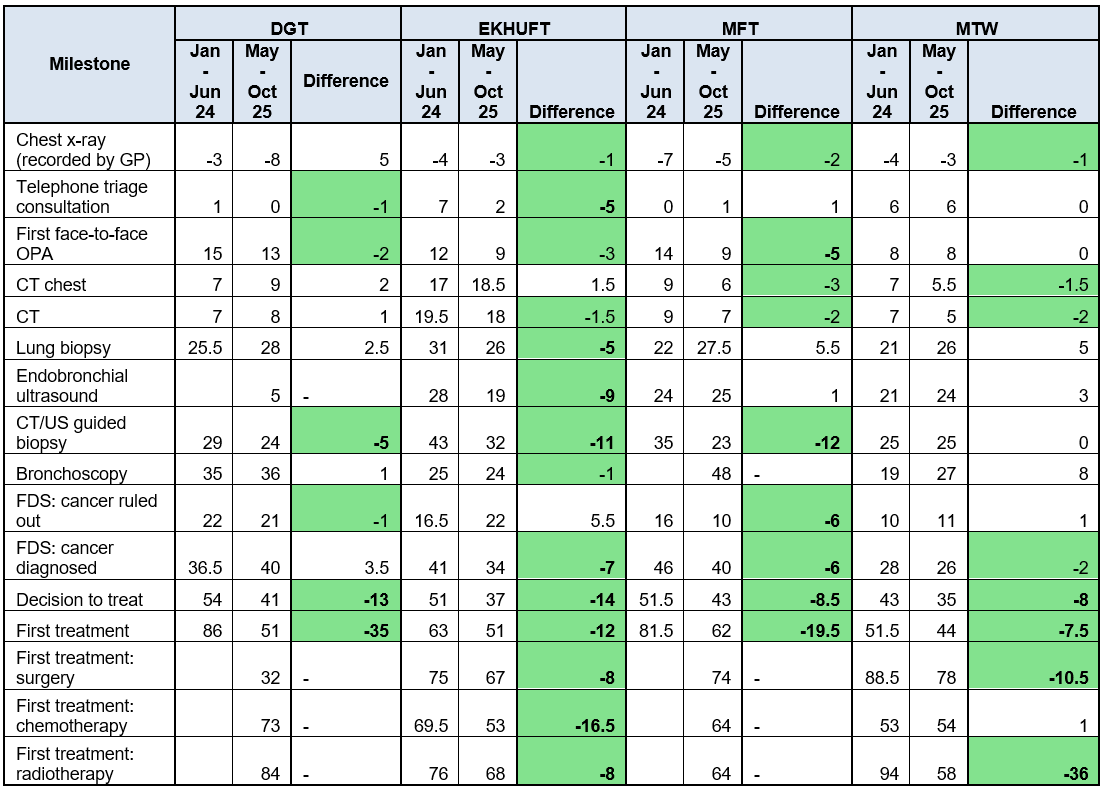
NHS Benefits
- Cancer Waiting Time Performance across the trusts have improved working towards the target thresholds (Table 2):
- Two trusts improved their 28-day FDS from baseline to evaluation, working towards reaching the 75% target. The other two trusts showed a decline but still met the 75% threshold.
- Improvements in 31-day CWT’s can be seen across 3 trusts achieving 100%, with one trust declining and missing the 96% target threshold.
- All four Trusts have improved 62-day CWT performance, from baseline to evaluation. Two Trusts have now met the 85% target threshold.
Table 2: Cancer Waiting Times Performance for Lung Pathway: Baseline April-June 2024 vs Evaluation July-September 2025
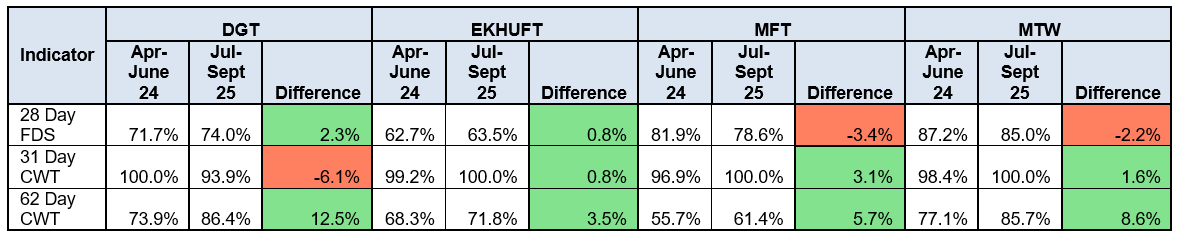
- The collaboration and networking across the trusts and across the Tumour Site Specific Group for the whole of Kent and Medway has improved because of this project, with multiple share and learn opportunities.
- The project identified long-term opportunities for the Cancer Alliance, which are now part of the Clinical Reference Group Work Plan for 2025/26. These include:
- Working with the Southeast Regional Cancer Alliances to develop a standard Inter Patient Transfer (ITP) Policy.
- Arranged better communication channels with our PET (Positron Emission Tomography) services to ease delays and improved the process to raise queries.
- Began work to investigate the regional Navigational Bronchoscopy and EBUS (Endobronchial Ultrasound) services – starting with the CRG surveying our operators.
- Improved tertiary surgical pathway with Guys & St Thomas Foundation Trust by improving.
- Worked to improve Radiology Data Quality utilising the CRG to liaise with the Trusts.
- Short term Trust level improvement plans were also developed and executed highlights include:
- CT scan waiting times reduced from 14 to 8 days at Medway (new target of 3 days); outpatient appointments now within 2 days of CT scans; East Kent offers hot reporting of chest X-ray reviews.
- Straight-to-Test (STT) pathway activated across East Kent cutting unnecessary steps; diagnostic test bundles implemented; automated cross-cover system to streamline MDT workload.
- Additional Lung Module MDM fully funded for integrated care; standardised referral and tracking planned; regional action sheets introduced to streamline oncology and radiotherapy bookings.
- Training additional respiratory consultants boosting EBUS capacity; CNS workforce expansion planned based on patient volume; adjusting job planning to improve consultant efficiency and MDT prep.
- Exploring one-stop clinics; improving senior nurse-led communication strategies enhancing engagement and reducing anxiety; CNS lead initiatives- chemotherapy chat clinics and breaking bad news support.
- Data collection and audits monitor delays in PET scans, biopsies, and molecular testing; age-based CT scan protocols (45–85) to optimise resources.
MSD Benefits
- Enhanced reputation and legitimacy for partnership work with MSD. Seven NHS stakeholders completed the survey following the PDP and results are shown below:
- Average score 4.6/5 and 3.8/5 was scored for being satisfied with MSD’s collaborative working project and believed MSD contributed to their organisation’s pathway respectively.
- Average score of 4.3/5 was scored for likelihood of working with MSD or the pharmaceutical industry in the future.
- Quotes taken from the survey when questioned, “what has improved in your service/pathway following this collaborative working project” from NHS Stakeholders:
- “Understanding the bottlenecks in our diagnostic pathway and identifying the drivers to change”
- “There have been many aspects of the pathway improvement – to added biopsy lists – better processes for PET CT, to patient engagement and attendance of tests. Also, better processes of bundling tests to get them in a more timely manner”
- When questioned on feedback of your experience of working in collaboration with MSD:
- “It has been very helpful working with MSD who were able to focus everyone on what the issues were through the workshops and then putting the onus back on the staff with some advice on how to make the necessary changes. Having the detailed lung pathway as it is currently mapped out by MSD was an eye opener and gave the team specific areas to focus on which could be quick wins to make improvements”
- “MSD was consistently engaged throughout the project, impartial in judgements, constructive in her comments and at times, candid about progress made. We really appreciated their expertise and motivation to help improve our cancer standards”
- This collaboration may have enabled more patients to access innovative treatments in line with NICE guidance which may or may not have included MSD medicines.
Funding & Resources
- This was a project with shared time commitment from Kent and Medway Cancer Alliance & MSD.
Lessons learnt
- Empowering the trust to take on this work has enabled improved progress in the CRG and TSSG. This ripple effect has meant there is an ease of collaboration for future programmes of work.
- It is critical to get a senior accountable person in each trust, and for them to have a space to report back the progress internally. E.g. we found that the clinical leads in Medway were able to deliver at pace.
- Taking the process mapping off-site definitely had the benefit of teams being able to focus and understand the importance of the case of change.
- The handover to the trusts could have been instigated earlier, as the project close felt like an abrupt stop.
Publications
- There are currently no plans to publish the outcomes of this project by the Cancer Alliance as per the date of this summary.
GB-NON-12771 | April 2026
Newcastle NHS Foundation Trust Lung Cancer Pathway Development Project (PDP)
Project Title
Newcastle NHS Foundation Trust Lung Cancer Pathway Development Project (PDP)
Organisations involved
MSD
Freeman Hospital
Royal Victoria Hospital
Summary
A National Optimal Lung Cancer Pathway (NOLCP) has been created with the intention of improving patient experience through promoting quality cancer care and ensuring all lung cancer patients receive optimal care. There was an opportunity in Newcastle to optimise day zero to day six of the NOLCP with a specific focus on reducing variation in the time from referral to first clinic appointment for those with radiology imaging showing suspicion of cancer. This ensured all patients, referred with radiology imaging showing suspicion of cancer, wait no longer than six days for their first clinic appointment. The project began on 1st March 2024 and finished on 28th February 2025.
Benefits Realised
Through implementation of MSD’s Pathway Development Programme with Newcastle Hospitals NHS Trust team, MSD provided project management support to assess the current state of the lung cancer pathway from day zero to day six, facilitated a gap analysis and worked with the Trust to develop an actionable implementation plan to deliver the changes required. As a result of the project, the following benefits were realised:
Patient Benefits
- A reduction of 3.75 days in the average time a patient with imaging suspicious of cancer was first seen in clinic, reducing average waiting time from 20.75 days to 17 days.
NHS Benefits
Improvement actions taken forward from this collaboration have optimised the lung cancer pathway, day zero to day six, across Newcastle Foundation Trust hospital sites resulting in the following benefits:
- All critical review notifications (CRNS) where imaging is suspicious of cancer is raised are now picked up and acted upon by Navigators who review twice weekly and send on to triage consultant to book to clinic.
- A chest x-ray optimal pathway for non-GP chest x-ray patients agreed with radiology and process now operational. Patients with imaging suspicious of cancer are now upgraded to optimal pathway, ensuring they are managed against optimal pathway time points. Patients upgraded onto the optimal pathway are those who are not referred for cancer but have an incidental finding on x-ray.
- From 2nd June 2025 the Trust is now able to flag upgraded optimal pathway patients on Somerset Cancer Register, which will allow them to run the data and interrogate with confidence how optimal patients are moving through the pathway aligned to best practice timed lung cancer pathway time points. As the system was only introduced in June a first data pull is not yet available to review impact of improvements.
- Triage process in progress to move non-cancer patients off the optimal pathway and into general respiratory clinic. Protocol now in place for patients with clear No Cancer to be directed to general respiratory clinic and Yes Cancer to cancer clinic. The Trust is now looking at formalising this triage process to reduce underutilisation of clinic slots.
- Prioritisation process for use of cancer clinic slots now supported by admin, with process to be formalised as a next step enabling the patients with imaging suspicious of cancer to be seen at the next available clinic slot.
- Nodule clinic funding was secured from Northern Cancer Alliance (NCA), and the service was launched on 2nd April 2025. Although this pathway change is in its early stages, the nodule clinic provides a way of managing patients outside of the cancer pathway for patients who do not have cancer but do require respiratory physician follow-up. This in turn frees up lung cancer clinic appointments for patients who do have cancer.
- Funding proposal submitted to NCA to recruit additional seven programmed activities (PAs) of consultant respiratory physician time at The Freeman. Awaiting outcome.
Early indication from June’s data is that the introduction of the nodule clinic has positively impacted 28 -day Cancer Waiting Times (CWTs), with compliance against faster diagnosis standard increasing to 85% against an average over the last two years of 78%. However, as these changes are in the early stages of implementation further data is required to determine whether this early indication of improvement is sustained.
MSD Benefits
- Increased understanding of the dynamics impacting the NOLCP from day zero to day six and the solutions to support patients with imaging suspicious of cancer to be seen in clinic by day six.
- Enhanced reputation and legitimacy for partnership work with MSD – Feedback received by Chris Ellis, Operational Service Manager – “This programme has been helpful in allowing us to work through the challenge in the data, gaining a greater depth of understanding of the early part of the pathway, and leading to tangible improvement actions. These things would not have been achieved without this collaboration”
- This collaboration may have enabled more patients to access innovative treatments in line with NICE guidance which may or may not have included MSD medicines.
Funding & Resources
This was a project with shared time commitment from Newcastle NHS Foundation Trust & MSD.
Lessons learnt
The importance of:
- Strong clinical leadership with a clear vision for change is necessary to effect change
- Identification of a Trust project manager resource to support implementation of the action plans from the end of the project through the immediate six-month period to evaluation.
- Trust internal stakeholder activation to prioritise pathway improvement activity
- Aligning on a communication plan for disseminating the incremental success of change during the project to maintain momentum.
Publications
No publications have been planned. There are currently no plans to publish the outcomes of this project by the hospital Trust as per the date of this Outcomes Summary.
GB-NON-11768 | August 2025
South Yorkshire and Bassetlaw Cancer Alliance Lung Cancer Pathway Development Project
Project Title
South Yorkshire and Bassetlaw Cancer Alliance Lung Cancer Pathway Development Project
Organisations involved
- MSD
- South Yorkshire and Bassetlaw Cancer Alliance (SYBCA)
- Barnsley Hospital Foundation Trust
- Sheffield Teaching Hospital NHS Foundation Trust
- The Rotherham NHS Foundation Trust
- Doncaster and Bassetlaw Teaching Hospitals Foundation Trust
- Chesterfield Royal Hospital Foundation Trust
Summary
A National Optimal Lung Cancer Pathway (NOLCP) has been created with the intention of improving quality of cancer care and ensuring all lung cancer patients receive optimal cancer care. There was an opportunity in SYBCA to optimise the lung cancer pathway from day 3 to day 21 in line with the NOLCP to deliver diagnosis in the least invasive way, address variation in care and optimise outcomes. The desired outcome of this project was to optimise the diagnostic pathway, reduce variation in care and secure greater compliance with the NOLCP milestones and strengthened performance related to Cancer Waiting Times (CWTs). The project ran for approximately 12 months from 1st February 2024 to 31st January 2025.
Benefits Realised
Through implementation of MSD’s Pathway Development Programme with South Yorkshire and Bassetlaw Cancer Alliance, MSD provided project management support to map the current NOLCP from day 3 to day 21, facilitated a gap analysis and worked with the Cancer Alliance and the five Trusts aligned to this region to develop actionable implementation plans to deliver the changes required to achieve the desired outputs.
Patient Benefits
The average number of days from referral to decision to treat (DTT) for patients entering the lung pathway is now 2.91 days earlier following delivery of the pathway development programme (Table 1). Average of DTT to first treatment date has increased by 0.14 days. The treatment pathway was out of scope for this programme, but when this data is included, patients are now moving through the full pathway on average 2.77 days earlier overall.
Table 1: Source: SYBCA timed pathways 62 days standard
| Baseline Jan to April 2024 | Jan to April 2025 | Days saved | |
| Average number of days from referral to first seen in clinic | 7.03 | 5.69 | 1.34 |
| Average number of days from first seen to decision to treat | 37.64 | 36.07 | 1.57 |
| Average number of days from decision to treat to treatment date | 13.07 | 13.21 | -0.14 |
| Total days | 57.74 | 54.97 | 2.77 |
NHS Benefits
Strengthened performance of 62-day CWT has been achieved (Table 2), with data from the baseline to evaluation showing an upward trend towards the target threshold of 85%. Overall performance against the FDS 28-day standard (Table 3) has however fallen from the baseline in 2024.
Table 2: Source: SYBCA 62 Day performance Monitoring – Tumour level
| Jan | Feb | March | April | |
| 2024 baseline | 56.6% | 58.9% | 59.1% | 64.3% |
| 2025 | 65.9% | 73.3% | 61.8% | 64.6% |
Table 3: Source: SYBCA provider level FDS 28-day data
| Jan/Feb/March/April | May | |
| 2024 baseline | 83.77% | 79.69% |
| 2025 | 74.35% | 77.48% |
The Pathway Development Programme identified both short-term trust actions that would optimise the local pathways but also highlighted systemic longer-term opportunities for the Cancer Alliance to address variation and optimise the lung pathway which have formed part of their overarching improvement plan for 2025/26. A treatment variation task and finish group were established and led by the Cancer Alliance to drive accountability for and delivery of local and system wide improvement actions.
At the point of publication there are three improvement actions in progress but not yet completed. First, the development of a system-wide networked provision for endobronchial ultrasound. This is under review with consideration now also being given to CT-guided biopsy. Second, with the Health Innovation Network (HIN) scoping how artificial intelligence can be embedded into the pathway to support identified radiology workforce capacity for X-ray and CT. Third, seeking a regional funding solution to ensure all trusts can sustain pathway navigator roles.
The Cancer Alliance has identified improvement actions which they are planning to progress to strengthen this pathway further across the next 6-12 months. These include ensuring GP direct access to chest X-ray embedded in all trusts across the lung pathway, embedding system wide standardisation of test bundles, ensuring point of care testing embedded in all trusts across the lung pathways and that ICE is set up and GPs are utilising this consistently.
Short term Trust level improvement plans to optimise local pathways were also developed and executed.
MSD Benefits
- Better understanding of lung cancer patient needs
- Enhanced reputation and legitimacy for partnership work with MSD. Four NHS stakeholders completed the survey following the PDP and results are shown below:
- Average score 9/10 and 8/10 was scored for being satisfied with MSD’s collaborative working project and believed MSD contributed to their organisation’s pathway respectively.
- Average score of 8/10 would recommend working in collaboration with MSD to others.
- Quotes taken from the survey when questioned, “what has improved in your service/pathway following this collaborative working project” from NHS Stakeholders:
“Better engagement. More traction and focus around the pathway improvement to secure greater efficiency and resilience. Identification of local and system level pathway improvement actions”
“There is a greater understanding of where there are bottlenecks in the pathway, it enabled staff to understand the process as a whole as well as identify differences in the pathway between the trusts in the system”
- When questioned on feedback of your experience of working in collaboration with MSD:
“It was really beneficial to have someone impartial involved in the process of mapping, particularly to ask questions that might be overlooked by those involved in the pathway more heavily”
“As an alliance, we have been fortunate to partner with MSD before and as always, the experience of working together has been excellent. The MSD team apply a very thorough approach and methodology and engage well with clinical teams and stakeholders. There is excellent communication throughout and ready escalation of any slippages. The outputs are clear and readily translatable into improvement plans and actions”
- This collaboration may have enabled more patients to access innovative treatments in line with NICE guidance which may or may not have included MSD medicines.
Funding & Resources
This was a project with shared time commitment from South Yorkshire and Bassetlaw Cancer Alliance, Barnsley Hospital Foundation Trust, Sheffield Teaching Hospital NHS Foundation Trust, The Rotherham NHS Foundation Trust, Doncaster and Bassetlaw Teaching Hospitals Foundation Trust, Chesterfield Royal Hospital Foundation Trust and MSD.
Lessons Learnt
- The value of having a Task and Finish group led by the Cancer Alliance to maintain communication, engagement and maintain accountability to deliver Trust identified improvement actions
- The importance of identifying a Trust project manager to facilitate stakeholder engagement with the process and implementation of the action plans.
- A longer period is required to make and measure significant service improvements.
- Recognition that many factors influence pathway efficiency and resilience, and not all of these factors were in scope for this project.
Publications
No publications are planned outside of this Outcomes Summary.
GB-NON-11759 | September 2025
St Bartholomew’s Hospital Equity of Access to Lung Cancer Treatment for Diverse Communities Pathway Development Programme (PDP)
Project Title
St Bartholomew’s Hospital Equity of Access to Lung Cancer Treatment for Diverse Communities Pathway Development Programme (PDP)
Organisations involved
MSD
Barts NHS Trust- St Bartholomew’s Hospital
Summary
There was an opportunity in Barts NHS Trust to optimise the Lung Cancer Patient Pathway. Barts Health NHS Trust serves one of the largest, most diverse populations in the UK1. Providing equitable cancer care, including access to and inclusion in clinical trials, to this culturally and socially diverse demographic of patients is a priority for the trust and hospital. The Lead Lung Cancer Clinician felt there was a lack of clarity on the existing lung cancer patient pathway, especially in relation to the different touch points the clinicians have with the patient, and the difference in approach of the clinicians to these touch points. The primary objective of this project was the optimisation of the lung cancer patient pathway, contributing towards an increase in the uptake of standard of care treatment and inclusion in clinical trials amongst patients from diverse communities.
To achieve the objective, the first method was to map and understand the current lung cancer patient pathway from delivering the diagnosis results (breaking bad news) to delivery of either standard treatment or inclusion into a clinical trial, with a specific focus on understanding the needs of patients from diverse communities- an innovative approach to traditional pathway mapping. This was achieved through pathway mapping workshops and one to one interviews with the healthcare professionals (HCPs) and other relevant decision makers who are responsible for the patient pathway. During these sessions the resources and existing support (for patients and clinicians) surrounding the conversation HCPs have with patients during the consultations was understood. A Gemba Walk (used in management practice where leaders observe work processes in their actual context to understand operations and identify areas for improvement – a workplace walk through) was adapted and implemented. This involved walking the patient pathway as a patient, attending dummy appointments to gather valuable insights to the patient journey, observing the process, understanding the challenges and identifying opportunities for improvement.
The second method was to use the Gemba Walk, Workshop findings and data to understand the challenges and obstacles of the patient pathway from a diverse patient perspective and to address some of the inefficiencies seen within the pathway and support further service improvements to help increase access to cancer treatments (including clinical trials) in diverse communities. This was achieved though GAP Analysis workshops and action planning through the NHS implementation phase of the project. This project forms part of a broader piece of work with St Bartholomew’s hospital to help support improvements in treatment access for diverse communities including uptake of clinical trials- where this project has helped to inform and direct the interventions required. The project ran from March 2024 to Feb 2025.
Benefits Realised
As a result of the project, the following benefits were realised:
Patient Benefits
- Patients are better supported to make informed decisions on their standard of care treatment or clinical trial inclusion irrespective of demographic – this was not measured as part of the project; however, it is anticipated the NHS benefits below will support the patients in their decision making.
- An increase in the proportionate representation of diverse populations across both uptake of standard of care treatment and inclusion in clinical trials, see NHS Benefits below.
NHS Benefits
- Having an increased understanding of the challenges and inefficiencies of the pathway and potential options to further support in optimising the pathway for diverse communities, has led to the following areas of improvement:
- Provided information on diagnostic tests and treatment in multiple languages
- Improved access to, and utilisation of, interpreters
- Improved patients understanding and navigation of the diagnostic pathway
- Encouraged patients to feel involved in their decisions making via additional resources around treatments
- This project has informed a broader piece of work with St Bartholomew’s hospital to help support further improvements in treatment access for diverse communities including uptake of clinical trials, this includes exploring opportunities in the following areas:
- Concise and accessible patient information delivered at the correct time in a patient’s pathway was identified as an unmet need therefore a patient information repository / delivery solution is being explored
- Support is required to help navigate patients through the diagnostic pathway and by understanding the needs and challenges of patients from diverse communities from this project a bespoke support package for these patients via a potential new job role is also being explored
- Uptake of standard of care treatment and inclusion in clinical trials amongst patients from diverse communities was measured within this project, however due to small patient numbers it is difficult to draw conclusions with this data.
- Project Baseline data (July 2023 – July 2024) showed 11 out of 555 (2%) diagnosed lung cancer patients declined active treatment. The patients who declined treatment came from the following ethnic communities:
- 1 patient from Black or Black British Communities declined treatment out of the 30 diagnosed (3.3%).
- 1 patient from Other Ethnic Group Communities declined treatment out of the 23 diagnosed (4.3%).
- 2 patients from Asian or Asian British Communities declined treatment out of the 74 diagnosed (2.7%).
- 7 patients from White Communities declined treatment out of the 393 diagnosed (1.8%).
- Post Project data (February 2025 – May 2025) showed a similar percentage of patients that declined treatment. 4 out of 188 (2.2%) diagnosed lung cancer patients declined active treatment. The patients who declined treatment all came from White Communities- 4 patients out of the 132 diagnosed patients (3.1%).
- Project Baseline data (July 2023 – July 2024) showed 11 out of 555 (2%) diagnosed lung cancer patients declined active treatment. The patients who declined treatment came from the following ethnic communities:
- A clear pathway is now mapped and understood in relation to how HCPs and patients from diverse communities interact with the system in lung cancer. This has supported St Bartholomew’s Hospital to develop, in collaboration with CRUK, a patient support tool to facilitate understanding and engagement of patients during the pathway. MSD had no role in the development of this patient support tool.
- The project promoted self-reflection for NHS individuals, the pathway is challenging to navigate and understand and that there is a need to improve the patient experience to achieve better engagement, participation and ultimately treatment
- The Gemba walk aspect of the project provided independent feedback on how ‘getting the small things right’ can make all the difference when implementing the suggestions. This has also helped to inform St Bartholomew’s hospital to develop a virtual hospital tour
MSD Benefits
- Better understanding of the lung cancer patient pathway and the impact when treating patients from diverse communities.
- Enhanced reputation of MSD through partnership work- 6 NHS stakeholders completed a survey following the PDP and the results are shown below:
- Average score of 9.5/10 were satisfied with MSD’s collaborative working project
- Average score of 9.17/10 believed MSD supported their organisations development or improved cancer service
- Average score of 7.5/10 to likely work with MSD and the pharmaceutical industry prior to the collaborative project which then changed to an average score of 9.67/10 in favour of collaboration following the project
- Average score of 10/10 would recommend working in collaboration with MSD to others
- Quotes taken from the survey when questioned, “what has improved in your service/pathway following this collaborative working project” from NHS Stakeholders:
- “Deeper understanding of the challenges patients may face when attending the lung cancer service, understanding of clinical correspondence they receive, and the gaps in information we provide. We have discussed ways in which we can address issues, including digital solutions”
- “This project has greatly improved my understanding of the patient journey, from diagnosis to treatment, allowing me to identify inefficiencies and explore more effective approaches. Collaborating with the multidisciplinary team has been invaluable, fostering better communication, shared learning, and a more cohesive approach to care. These changes have enhanced the overall quality and efficiency of our service, benefiting both patients and staff”
- “HCP gained understanding from patient perspective. Pathway mapping has improved local practice. Potential new post is generated for service improvement for EDI purpose”
- When questioned on feedback of your experience of working in collaboration with MSD:
- “It has been a pleasure to work with Hannah and her team. The meetings have been appropriate length and frequency, friendly atmosphere creates open discussion, prompt and finish on time, excellent guidance during discussions and clear follow up emails with good summaries of discussion and action plan”.
- “I have had the opportunity to collaborate with MSD on various projects in the past, and my experience has always been very positive. The teams at MSD are highly professional, collaborative, and supportive, which has made working together both productive and enjoyable. Their commitment to innovation and patient-centred outcomes aligns well with my own values, and I’ve always appreciated their clear communication and willingness to listen to feedback. This project has been no exception“.
- “Organised, proactive and engaging to deliver this project”.
- This collaboration may have enabled more patients to access innovative treatments (including appropriate clinical trials) in line with NICE guidance which may or may not have included MSD medicines
Funding & Resources
This was a project with shared time commitment from St Barts NHS & MSD.
Lessons Learnt
- A better understanding of the improvement pathways within NHS trusts so that future MSD Collaborative Working projects may align better to NHS Quality-Improvement methodology and tools
- Regular touch points to ensure clarity of expectation and outcomes and a clear plan of action and review within stakeholder meetings
- The NHS teams became more cohesive during the project and as their understanding of the challenges their patients face, began to develop a coalition of willing and engaged individuals developed who wanted to make changes to the pathway to enhance its patient-centricity
Publications
There are currently no plans to publish the outcomes of this project by the hospital trust as per the date of this summary.
References
- NHS Choices, WeBelong – Barts Health NHS Trust (www.bartshealth.nhs.uk/webelong/), last accessed 26/06/2025
GB-NON-11628 | July 2025
Northern Cancer Alliance (NCA), Lung Cancer Getting It Right First Time (GIRFT) Implementation Project
Project Title
Northern Cancer Alliance (NCA), Lung Cancer Getting It Right First Time (GIRFT) Implementation Project
Organisations involved
NCA
MSD UK
Summary
This was a collaborative working project lasting 18 months between MSD UK and the NCA which completed in December 2024. NCA is responsible for overseeing the implementation of selected treatment recommendations from the national Lung Getting It Right First Time (GIRFT) report. The NCA has also identified improvement of Systemic Anti-Cancer Treatment (SACT) rates, introducing a Stage 3a Multi-Disciplinary Team (MDT) meeting and Inter-Patient Transfer (IPT) rules as priorities.
This project intended to work with the NHS Foundation Hospital Trusts (FTs) in the NCA geography (North Cumbria NHS FT, Northumbria Healthcare NHS FT, Newcastle upon Tyne Hospital FT, Gateshead NHS FT, South Tyneside and Sunderland NHS FT, North Tees and Hartlepool NHS FT, Country Durham and Darlington NHS FT and South Tees NHS FT) to implement the 3 selected, and reportable to NHSE GIRFT recommendations alongside improvement in SACT treatment rates for advanced lung cancer patients. and to implement a new Stage IIIa MDT (multi-disciplinary team) meeting for lung cancer and create and implement new IPT rules across the region.
Benefits Realised
Patients
- 11.4% more patients with Non-Small Cell Lung Cancer (NSCLC) stage IIIa are being treated with radical intent.
- 5.3% more patients are receiving radical intent treatment in a timely manner thus providing more consistent delivery of optimal care.
- Trust lung pathway navigators provide a single point of contact to guide the patient through a complex pathway.
NHS
- After mapping out the regional patient numbers identified with a NSCLC diagnosis of stage IIIa, it was decided that a regional Stage IIIa MDT was not operationally justifiable due to small patient numbers. It was later decided to move this to a regional stage III MDT which is currently in development by the Lung Pathway Board.
- Funding was secured for 8 Lung Pathway Navigator roles across all 8 trusts, most of whom were recruited prior to the project ending.
- Regional IPT form was developed and currently being trialled in Gateshead Hospital.
- GIRFT recommendation 9 – Radical treatment rate for patients with NSCLC, stage I-II, Performance status (PS0-2) was consistently maintained above the 85% GIRFT target.1
- GIRFT recommendation 15 – Trusts should record and monitor multimodality treatment in NSCLC stage IIIa disease and offer radical intent treatment as standard in fit patients improved from 79% to 90.4%.2
- GIRFT recommendation 16a – NSCLC, radical intent treatment should commence by day 49 of the overall National Optimal Lung cancer Pathway (NOLCP). This improved from 6.3% to 11.6%.3
- GIRFT recommendation 16b – NSCLC radical intent treatment to start by day 16 post decision to treat (DTT) in line with the NOLCP. This improved from 15.4% to 36.3%.4
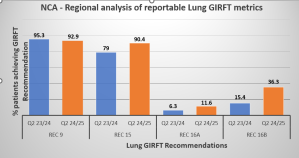
- The use of SACT to treat NSCLC, stage IIIb – IV, PS 0-1 showed a decline over the duration of the project of 73.3% to 53.4%.5 Whilst no reason was attributable to this decline in SACT treatment rates it could be due to a number of factors i.e. Patient choice, declining performance status.
- 62-day Cancer Wait Time (CWT) data for the lung pathway has improved from 52% (May 2023) to 63.7% (December 2024). Moreover, the region is now achieving above the national average of 61.8% for the 62-day lung CWT and has moved from the lower middle 25% of ranked Integrated Care Boards (ICBs) to the upper middle 25% of ranked ICBs.6 There has also been a 25% increase in the number of patients per month receiving treatment from an average of 200 per month in 2023, to 250 patients per month in 2024.
- 31-day CWT has shown a general upward trend throughout a 13-month period – starting from 90% in June 2023 and improving to 96.7% in June 2024 where it maintained around 95%. This improvement was due to an increase in theatre capacity for lung surgery, and recruitment of a substantive cardiothoracic surgeon. This enabled surgery on weekends. From September 2024 – December 2024 the 31-day CWT dropped to around 85%7, when radiotherapy encountered capacity issues due to one of the LINAC machines being out of service.
MSD
- Greater understanding of the pressures within the lung pathway in relation to treatment, and the fragility of services especially when equipment fails to work i.e. linac radiotherapy machine.
- Enhanced reputation of MSD through partnership work.
- This collaboration may have enabled more patients to access innovative treatments in line with NICE guidance which may or may not have included MSD medicines.
Funding & Resources
This was a project with shared time commitment from Northern Cancer Alliance & MSD UK. No funding was associated with this project.
Lessons learnt
Due to the dynamic nature of new treatment approvals, and ever-changing treatment options within the lung pathway, the ability to implement GIRFT recommendations at speed is paramount.
Although only 3 GIRFT metrics needed to be reported to NHSE, NCA wanted to include another GIRFT indicator to be monitored/improved which was deemed important to reducing treatment variation for advanced NSCLC patients.
Publications
To date, there are currently no plans to publish the outcomes of this project by the NCA outside of this Outcomes Summary.
References
1 Data provided by local trusts data analytics department.
2 Data provided by local trusts data analytics department.
3 Data provided by local trusts data analytics department.
4 Data provided by local trusts data analytics department.
5 Data provided by local trusts data analytics department.
6 Data provided by Northern Cancer Alliance business insights department.
7 Data provided by Northern Cancer Alliance business insights department.
GB-NON-11553 | July 2025
NHS Tayside Lung Cancer Pathway Development Project (PDP)
Project Title
NHS Tayside Lung Cancer Pathway Development Project (PDP)
Organisations involved
MSD
NHS Tayside
Summary
The Scottish National Optimal Lung Cancer Pathway (SNOLCP) has been created with the intention of improving patient experience through promoting quality cancer care and ensuring all lung cancer patients receive optimal cancer care. There was an opportunity in NHS Tayside to optimise the lung cancer pathway in line with the national optimal lung cancer pathway and to improve the service quality, service efficiency, productivity and patient experience. The desired outcome of this project was an improved lung cancer pathway aligned with the SNOLCP and an improvement in the cancer waiting time targets. The project ran for approximately 15 months.
Benefits Realised
Due to significant and unanticipated capacity issues within NHS Tayside which needed to take priority, the project did not complete in the pre-defined timescales, and therefore no benefits were realised as a result.
This collaboration may have enabled more patients to access innovative treatments in line with SMC guidance which may or may not have included MSD medicines.
Funding & Resources
This was a project with shared time commitment from NHS Tayside & MSD.
Lessons learnt
Creation of a formal contingency plan should any of the key stakeholders and MSD project manager have time capacity issues or their organisation suffers significant disruption which affects stakeholder capacity during the implementation of the project.
Publications
There are no intended publication plans outside of this Outcomes Summary.
GB-NON-11566 | June 2025
Manchester University NHS Foundation Trust Patient Experience App
Project Title
Manchester University NHS Foundation Trust Patient Experience App
Organisations Involved
Manchester University NHS Foundation Trust, MSD
Summary
Wythenshawe Hospital (Manchester University NHS Foundation Trust) have initiated a RAPID (rapid access to pulmonary intervention and diagnosis) pathway for all lung cancer patients. It aimed to reduce the time from GP referral to first treatment to 28 days, less than half the current NHS target. While the pathway had demonstrated advantages in time to diagnosis, further evidence was needed to understand the impact of such a fast-track diagnosis on the patient experience. The aim of the project was to work with Manchester University NHS Foundation Trust to develop a patient experience survey. The purpose of the survey was to build an evidence base of what the experience of the patient is within this accelerated pathway, and to facilitate continuous improvement to the pathway through real-time analysis.
Benefits Realised1
The survey included a total of 322 participants, comprising of 283 patients and 39 family/carers. 50% of survey respondents were over 70 years old, 20% were from the most deprived areas, and 96% identified as ‘White’ ethnicity, prompting reflection on whether this reflects the served communities.

Over 200 positive comments were received, praising the care received at Wythenshawe, while approximately 40 negative comments highlighted issues such as waiting for CT results and negative experiences in the RAPID hub waiting area.
Safe-7: Seven questions were asked around safety across the following categories: speed, communication, accessibility, kindness, trust, listening and overall safety. All categories demonstrated improvements from the baseline score with final scores of 9 and above, out of 10. The overall safety category rated was 9.6 out of 10.
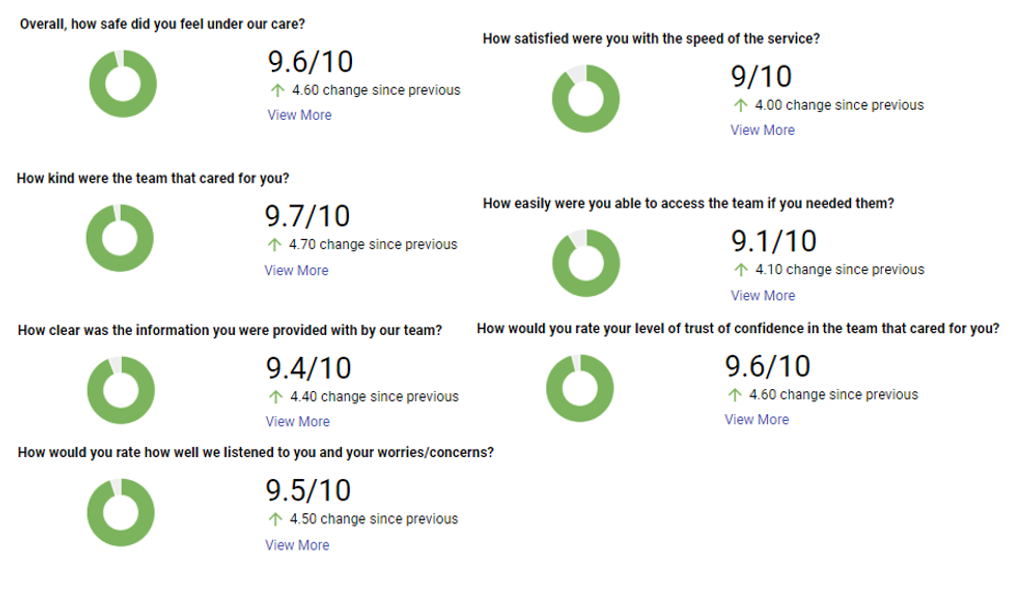
Benefits to patients:
- This project incorporated patient feedback to enhance the RAPID pathway, although due to insufficient data, some areas of improvement were not fully measured. The app provided insights into thoracic oncology services, assessing if high-quality care was being provided.
- The patients involved were grateful for the integration of Safe-7 into practice.
- There is a request to provide Safe-7 in the top 5 non-English languages in Greater Manchester and in an easy-read format to ensure equitable access.
Benefits to NHS:
- Capturing this patient experience data has enabled the Trust to demonstrate the pathway’s performance and deliver effectively.
- The report acknowledges the need for development and reconfiguration of clinic templates.
- The report encourages the continued integration of Safe-7 into daily practice and invites ideas for enhancements and opportunities for conference abstracts and staff publications.
- There are now plans of scaling up the project to expand beyond Manchester Foundation Trusts to other trusts across Greater Manchester and across other cancer sites.
Benefits to MSD:
Data generated on the patient pathway and experience prior to treatment decision could be used by MSD for:
- Policy and advocacy activities
- Development of patient support materials
- Advocacy for the RAPID pathway model
- Collaborating on this high-profile, innovative project has enhanced our reputation, leading to opportunities for further expansion across Greater Manchester Trusts.
This collaboration may have enabled more patients to access innovative treatments in line with NICE guidance which may or may not have included MSD medicines.
Funding & Resources
This project was a shared funding commitment from Manchester University NHS Foundation Trust & MSD. The total project cost was £79,150.
Lessons learnt and possible project expansion:
Languages
The Safe-7 survey report shows that 96% of respondents are of ‘White’ ethnicity, indicating the need to make the survey more accessible and equitable. To achieve this, the survey will be made available in the following languages: Arabic, Bengali, Cantonese, Easy Read, Farsi, Gujarati, Polish, Punjabi, Romanian, and Urdu. The optimal implementation approach would involve linking the invitation to a specific language and providing a language-switching option on the survey platform to ensure inclusivity.
Selecting the Service Area within the Email Invitation
The current practice of asking participants to select the service area on the survey’s home page has been found to be error-prone and overly technical for laypersons. The proposed solution is to allow participants to select the service area within the email invitation, providing a more user-friendly experience and reducing potential errors in service area selection.
Completing the Survey without an Email Invitation
To increase the survey’s uptake, there is a proposal to enable patients to complete the survey directly, without needing an email invitation. This additional option aims to accommodate patients who prefer to complete the survey during clinic consultations. The new process would include the ability to select the service area and language and could be accessed directly via a phone or provided iPad, in addition to the traditional email invitation.
New Service Areas Expansion of the Safe-7 project to Manchester Royal Infirmary and North Manchester General Hospital will require adjustments to the email communication, replacing references to Wythenshawe Hospital with ‘Manchester University NHS Foundation Trust’.
Publications
The system now has >1500 responses, The Christie have commissioned an independent analysis of the outcomes which will be planned for formal publication.
References
- MFT Safe7 Report October 2023
GB-NON-10332 | October 2024
Hywel Dda University Health Board lung Pathway Development Project (PDP)
Project Title
Hywel Dda University Health Board lung Pathway Development Project (PDP)
Organisations involved
MSD
Hywel Dda University Health Board
Summary
A national optimal lung cancer pathway has been created with the intention of improving patient experience through promoting quality cancer care and ensuring all lung cancer patients receive optimal cancer care. There is an opportunity in Hywel Dda University Health Board (HDUHB) to optimise the lung cancer pathway in line with the national optimal lung cancer pathway to improve the service quality, service efficiency, productivity, and patient experience. The desired outcome of this project is an improved lung cancer pathway aligned with the national optimal pathway and achievement of the lung cancer 62 day Cancer Waiting Time Targets. The project intended to start on 1st April 2022 and was extended to finish on 31st December 2023.
Benefits Realised
Due to significant and unanticipated capacity issues within the HDUHB system which needed to take priority, it has not been possible to complete the outcome summary for the HDUHB lung cancer pathway PDP.
Funding & Resources
This was a project with shared time commitment from HDUHB & MSD
Lessons Learnt
Creation of a formal contingency plan should any of the key stakeholders and MSD project manager have time capacity issues or their organisation suffers significant disruption which affects stakeholder capacity during the implementation of the project.
Publications
No publications are planned.
GB-NON-09771 | July 2024
EPIC: Early Prehabilitation in Lung Cancer
Project Title
EPIC: Early Prehabilitation in Lung Cancer
Organisations involved
NHS Lothian, Edinburgh Cancer Center, South East Scotland Cancer Network (SCAN) & MSD
Summary
The Edinburgh Cancer Centre, NHS Lothian and SCAN worked with MSD to understand the feasibility of utilising prehabilitation techniques in patients with advanced metastatic lung cancer with the overall aim of reducing symptom burden, improving patient fitness and increasing treatment rates for lung cancer.
Lung cancer is the most common cancer in Scotland with more than 5,500 registrations in 20191 and it accounted for over 25%2 of all cancer deaths. Around 50% of patients are identified at stage 41 meaning that access to treatment can be limited and this is compounded by the fact that many patients have comorbidities and are frail which adds to the symptom burden3.
Prehabilitation is the practice of enhancing a patient’s functional and psychological capacity before treatment commences. Ideally, prehabilitation interventions start at diagnosis, helping people to prepare for the next treatment stage in their journey of care.
The project was based at St John’s Hospital, Livingston and aimed to include all patients with locally advanced and advanced lung cancer (visible metastases or mediastinal nodes on diagnostic CT scan). It was run over 30 months including during the covid pandemic which impacted the expected patient numbers.
The principal objectives of the project were to –
- maximise patient fitness as they are investigated and start treatment for lung cancer
- Reduce symptom burden
- Improve nutrition and stop weight loss
- Increase treatment rates for lung cancer
Benefits Realised
Patients
- Patients may be more likely to access treatment and the project may improve their overall survival versus a matched cohort
- Patients are less likely to attend A&E, be admitted to hospital and if admitted spend less time in hospital versus a matched cohort
- Patients nutritional needs are better understood and their weight is managed more effectively
- Adaptations made to the prehabilitation program between phase 1 and phase 2 of the project improved patient uptake by managing patient symptoms before accessing physiotherapy and nutritional input
- 93% of patients who participated in the project said that they “would recommend others to attend” the prehabilitation clinic
NHS Benefits
- Implementing a prehabilitation service within the existing lung cancer pathway signals improved outcomes for advanced lung cancer patients and contributes to meeting NHS Lothian’s objectives
- Patients enrolled in the prehabilitation program experience reduced A&E admissions (20.4 adm’s vs. 28.3 adm’s), inpatient hospital admissions (51.9 adm’s vs. 67.9 adm’s), and shorter hospital stays (2.5 days vs. 8 days) compared to a matched cohort, helping to alleviate capacity pressures and freeing up vital resources to support other patients
- Scaling the prehabilitation service across NHS Scotland has the potential to unlock over 10,000 bed days per year, presenting a substantial opportunity to enhance capacity and resource allocation
- The EPIC project closely aligns with the goals and action points of the Scottish Government’s Cancer Strategy and Action Plan, particularly its focus on lung cancer and the urgent need for comprehensive action and resource allocation to address the complex challenges associated with this disease, making prehabilitation initiatives a valuable support approach
MSD Benefits
- Enhanced reputation of MSD through partnership work. NHS stakeholders completed a survey following the project and the results are shown below:
- Average score of 8.5/10 was scored for being satisfied with MSD’s collaborative working project and 7.8/10 believed MSD contributed to their organisation’s cancer service pathway respectively.
- Average score of 7.5/10 would recommend working in collaboration with MSD to others.
- Quotes taken from the survey when questioned, “what has improved in your service/pathway following this project–
- “Engagement between health care professionals, charitable services and patient community. Valuable in building evidence base on prehabilitation and how it can be integrated into existing pathways.”
- When questioned on feedback of your experience of working in collaboration with MSD –
- “Been valuable to be involved. Allocating time to such project development can be difficult given competing demands. I think this project has enhanced cross-team working and opportunities to share good practice.”
- “It has been helpful to have MSD drive some of the project forward – this takes some pressure off clinicians”
- Increased understanding of the lung cancer pathway, the challenges to current service provision and potential solutions to support patients.
- This collaboration may have enabled more patients to access innovative treatments in line with SMC guidance which may or may not have included MSD medicines.
Funding & Resources
This was a project with shared time commitment from NHS Lothian & MSD
Publications
- Early prehabilitation in suspected locally advanced and metastatic lung cancer (https://spcare.bmj.com/content/13/e3/e908)
- Early prehabilitation reduces admissions and time in hospital in patients with newly diagnosed lung cancer (https://spcare.bmj.com/content/early/2024/04/17/spcare-2024-004869)
References
- Cancer incidence in Scotland – Cancer incidence and prevalence in Scotland to December 2019 – Cancer incidence in Scotland – Publications – Public Health Scotland (https://publichealthscotland.scot/publications/cancer-incidence-in-scotland/cancer-incidence-in-scotland-cancer-incidence-and-prevalence-in-scotland-to-december-2019/)
- Scottish – ScotPHO (https://www.scotpho.org.uk/health-conditions/cancer-lung/data/scottish/
- The prevalence of comorbidity in the lung cancer screening population: A systematic review and meta-analysis – PubMed (nih.gov)
GB-NON-09668 | June 2024
Pan Tumour – Active Projects
Cancer Diagnostic Pathways in Community Diagnostics Centres
Project Title
Cancer Diagnostic Pathways in Community Diagnostics Centres
Organisations involved
Calderdale and Huddersfield NHS Foundation Trust
Mid Yorkshire Teaching Hospital NHS Trust
MSD UK
Summary
In 2025 the Labour government published a new 10-year health plan (1). Within this plan was: moving from hospital to community. Calderdale and Huddersfield NHS Foundation Trust, and Mid Yorkshire Teaching NHS trust, are now working towards service transformation for cancer patients and moving diagnostics out into the community for the following cancer pathways: Head & Neck, Lung, Gynaecology, Upper Gastrointestinal (UGI). The project will be collaborative working between Calderdale and Huddersfield NHS Foundation Trusts, Mid Yorkshire NHS teaching Trust and MSD UK, for a duration of 12 months.
Background
Calderdale, Kirklees, and Wakefield (CKW) serve a diverse population with varying cancer outcomes due to differences in referral routes, diagnostic access, and workforce structures. This leads to inconsistent diagnosis times, repeated tests, and varied patient experiences. Head and neck cancer patients often face multiple appointments across sites, causing delays and added stress. Community Diagnostic Centres (CDCs) offer a chance to streamline these pathways, but no standard model exists for the four main cancer types. This project aims to create and pilot a unified, efficient diagnostic model across CKW, supporting regional goals for faster, fairer cancer diagnosis and improved patient care in the following cancer pathways: Head & Neck, Lung, Gynaecological, Upper gastrointestinal (UG). This work aligns with existing regional priorities around cancer waiting times, early diagnosis, health inequalities, and system productivity, while supporting more consistent, patient-centred care delivery across West Yorkshire.
.
Project Objectives
- Regional standardisation of diagnostic referral pathways to CDCs for suspected cancer.
- Attainment of national Best Practice Timed Pathways (BPTP).
- Alignment with the recently published 10-year NHS plan – from hospital to community
- Capacity releasing of diagnostic services in acute care settings
Benefits
Patients
- Reduction in secondary care visits during the diagnostic phase of the patient pathway
- Reduction in patient uncertainty and anxiety by providing faster diagnosis
- Quicker time to treatment
NHS partner
- Increased diagnostic efficiency across the region
- Diagnostic capacity releasing in secondary care
- Strengthened collaboration between acute trusts, primary care and CDCs
- Reduction in variation in the 4 cancer diagnostic pathways across the region
MSD
- Reputational benefit of MSD working with the NHS to improve services
- As a pharmaceutical manufacturer of oncology medicines, an indirect result could be that MSD may see use of their NICE approved medicines
- An improved understanding of how care can be moved into the community to alleviate acute care operation pressures
Funding & Resources
This project is a shared contribution of time between Calderdale & Huddersfield NHS Foundation Trust, Mid Yorkshire Teaching Hospitals NHS Trust and MSD.
GB-NON-12732 | April 2026
Royal Cornwall Hospital Remote Patient Monitoring of ePROMS
Project Title
Royal Cornwall Hospital Remote Patient Monitoring of ePROMS
Organisations involved
Royal Cornwall Hospitals NHS Trust, Technical Health Ltd (MySunrise), MSD UK
Summary
This collaborative project aims to implement and evaluate a remote patient monitoring (RPM) service for Systemic Anti-Cancer Therapy (SACT) patients at Royal Cornwall Hospitals NHS Trust using the MySunrise App. The service enables patients to complete digital wellness questionnaires and report symptoms, prompting earlier engagement with Acute Oncology Services (AOS). The project will help redesign cancer pathways, reduce pressure on oncology services, enhance patient self-management, and support earlier detection of issues to reduce avoidable emergency presentations. Implementation and evaluation activities will run over 12 months, aiming to contribute to capacity release, improved patient experience, and development of a repeatable implementation playbook.
Background
SACT activity is increasing nationally by 6–8% annually,1 driving demand on oncology and acute services. Rising patient volumes, workforce shortages, and growing complexity of treatments mean many patients experience unreported or undetected symptoms that escalate into emergency admissions. Remote monitoring offers a mechanism to support timely intervention, improve patient experience, and mitigate pressures across AOS services. The MySunrise platform provides accessible, multilingual, mobile-enabled digital tools to support patient activation and symptom reporting during treatment.
Project Objectives
- Patients:
- Improve clinical monitoring and support, potentially improving outcomes.
- Enable patients to self-monitor symptoms and report side effects earlier.
- Strengthen connectivity between patients and clinical teams.
- Support out-of-hospital management and reduce unnecessary travel burdens.
- NHS:
- Reduce demand on oncology and acute services through proactive digital monitoring.
- Release clinic capacity and improve workflow efficiency for AOS and SACT nurses.
- Reduce hospital admissions and associated episode costs in the long term.
- Enable pathway redesign and modernisation of cancer services.
- MSD:
- Contribute to significant healthcare innovation that improves patient care and service efficiency.
- Strengthen partnerships with NHS organisations and enhance reputation.
- Improve internal capability in developing robust evaluation approaches.
- Enable scalability of RPM approaches to other settings and Trusts.
Benefits
- Patients:
- Earlier detection and support for SACT-related symptoms.
- Improved experience through reduced travel and convenient clinical contact.
- Increased confidence, engagement, and empowerment.
- NHS:
- More efficient resource use and potential reduction in hospital admissions.
- Improved adherence, satisfaction, and fewer missed appointments.
- Pathway efficiencies and release of clinical capacity.
- MSD:
- Advancement of digital innovation in cancer services.
- Strengthened NHS collaborations.
- Development of scalable evaluation frameworks.
Funding & Resources
This project is a shared contribution between Royal Cornwall Hospitals NHS Trust, Technical Health Ltd, and MSD The total project cost is £60,000.
GB-NON-12540 | March 2026
NHS Wales Digital Patient Alert Cards
Project Title
NHS Wales Digital Patient Alert Cards
Organisations Involved
NHS Performance and Improvement (Public Health Wales) and Merck Sharp and Dohme (UK) Limited
Summary
The purpose of this project is to digitise the standardised paper-based cancer patient alert cards across NHS Wales. All Wales cancer treatment alert cards have been in use since 2015 with the aim of providing a succinct and compact reminder for patients of the key symptoms to report, the 24-hour helpline number to call and key information for healthcare professionals who may be involved in their care. The paper-based card system will remain in place, and a QR code added which will link to the digital alert card, giving patients options regarding which system suits them best and providing a failsafe. The information contained on the current paper-based card will be used within the digital format. Evaluation of the digital and paper-based alert card options will be used to optimise the alert card systems going forward.
Background
Currently, patients receiving Systemic Anti-Cancer Therapy (SACT) are given a fold out paper alert card which should be carried by them and presented to any healthcare professionals (HCPs) who they may need to inform about their current treatment regimen. The card helps associated HCPs to understand the differences between the types of SACT that a patient may be receiving and also what associated medical care to give, not give (contra-indications) and also when to refer to specialists. Evaluation of the paper-based alert cards have identified potential improvements with patients and healthcare professionals reporting that physical cards are often lost or simply forgotten when patients have contact with the wider healthcare system, particularly when new or alarming symptoms occur. A digital version of the card that sits within a patient’s digital wallet on their smartphone, or with their permission, on the device of a family member/care-giver, will enable patients to provide the wider healthcare community with information about the type of SACT they are receiving if the physical card is lost or forgotten, or if they prefer a digital version. Patients will be able to access their alert card from their digital wallet without data or Wi-Fi.
Project Objectives
- Increase the use of patient alert cards by patients to inform other HCPs about the type of treatment they are receiving for their cancer care.
- Provide allied HCPs with information (that was previously contained on the paper-based version of the alert card) surrounding patients’ cancer care to allow them to treat associated or non-associated medical conditions appropriately and safely.
Benefits
Benefits to Patients
- A way of carrying important information concerning their SACT that can be accessed
- An improved patient experience when accessing healthcare not directly associated with oncology
Benefits to the NHS partner
- HCPs not directly associated with oncology services will have greater access to information surrounding a patient’s SACT through the digital alert card.
- This information will help to educate HCPs around SACT, the different types of SACT and how patients should be treated if other medical conditions occur.
- Potential efficiencies based on increased knowledge which may result in patients staying on their intended therapy for as long as possible within the recommended treatment regimen.
Benefits to MSD
- Enhanced reputation of MSD through partnership
- Patients receiving SACT including immunotherapy will be managed more efficiently by the wider HCP community. This may result in patients staying on their respective therapy due to enhanced knowledge around interactions and potential side effects.
Funding & Resources
This project is a shared contribution between NHS Performance and Improvement (Public Health Wales) and MSD. The total project cost is £10,200.
GB-NON-12380 | December 2025
ANP-SACT: Advanced Nurse Practitioner Led – Systemic Anti-Cancer Therapy Project
Project Title
ANP-SACT: Advanced Nurse Practitioner Led – Systemic Anti-Cancer Therapy Project
Organisations involved
NHS Lothian & MSD
Summary
South East Scotland Cancer Network (SCAN) serves a population of over 1.5 million people and, like many cancer networks, faces significant capacity challenges due to rising patient numbers and a shortage of clinical staff. This project aims to assess the service implications of delivering outpatient SACT (Systemic Anti-Cancer Treatment) through an Advanced Nurse Practitioner (ANP) led model rather than the traditional consultant-led approach. It will also evaluate the impact on overall capacity, particularly the potential benefits of freeing up consultant oncologist time for other critical areas of care. Based on the findings of this evaluation, consideration will be given to whether this model could be scaled and implemented across other hospitals and health boards in Scotland. The project will run for approximately 18 months.
Background
The project is needed due to a critical shortage of oncologists and radiologists in Scotland, with deficits projected to worsen by 2029, prompting the need for innovative care models. Advanced Nurse Practitioners (ANPs) can help address this gap by leading clinics that offer faster, more flexible access to care, especially for working patients and those with family responsibilities. Their close collaboration with Multidisciplinary Teams ensures coordinated and consistent care, while their role in patient education and emotional support enhances treatment adherence. ANP-led services are also more cost-effective, yield high patient satisfaction, and support the Scottish Government’s Cancer Strategy 2023–33 by promoting equity and expanding healthcare capacity.
Project Objectives
The project will aim to the measure and deliver on the following, using data from current services with consultant led and delivered clinics as a baseline from which to compare-
- The number of patients managed by ANP led clinics and no longer being seen in consultant-led clinics
- Inpatient and emergency admissions and days in hospital
- Treatment delays
- Number of patients stopping treatment early
- Staffing cost per patient
- Patient experience of nurse led service via a questionnaire – the focus will be on patient satisfaction with the experience with the clinic, no patient reported outcomes (PROs) related to patients’ health-related quality of life will be collected
- HCP experience of nurse led service via a questionnaire
Benefits
Benefits/ Impact to patients
- Potential improved patient experience
- Increased service efficiencies can lead to quicker access to appropriate treatment
- Responsive service to promptly manage questions or new problems
Benefits to the NHS partner
- Potentially scalable project that could be adopted by other Health Boards/Scottish Government
- Expansion of existing skill base in the nursing services in NHS Lothian
- Potentially improved capacity and costs savings for NHS Lothian
- Evaluation of novel service model
- Potential freeing up of consultant oncologist time to focus on more complex cases
Benefits to MSD
- Opportunity to engage as a partner with NHS Lothian and SCAN and to expand the knowledge of service design and patient throughput
- Better understanding of the patient journey through the non-metastatic cancer pathway
- Enhanced reputation of MSD through partnership work
- As a pharmaceutical manufacturer of oncology medicines, an indirect result of an improved pathway may be that MSD see more usage of their SMC reimbursed medicines
Funding & Resources
This project is a shared contribution between NHS Lothian and MSD. The total project cost is £80,889.
GB-NON-12198 | November 2025
Immuno-oncology Nurse led Regional Service in Humber Health Partnership
Project Title
Immuno-oncology Nurse led Regional Service in Humber Health Partnership
Organisations involved
MSD UK
Humber Health Partnership
Humber and North Yorkshire Cancer Alliance
Summary
Humber Health Partnership (HHP) is one of the largest groups within the NHS, made up of 2 trusts – Hull University Teaching Hospitals NHS Trust (HUTH) and Northern Lincolnshire and Goole NHS Foundation Trust (NLAG) serving a population of 1.65 million people.
MSD UK have been approached by Humber Health Partnership to support the set up and development of an Immuno-oncology nurse led service, with the emphasis on patients receiving checkpoint inhibitors (CPIs). This project is intended to run for 24 months.
Background
Immunotherapy has significantly advanced cancer treatment over the past decade, although it introduces some complexities and uncertainties regarding its administration and mechanism of action.
Across the trusts within Humber Health Partnership there is variation as to how adverse events for patients receiving CPIs are managed. This project will support the regional standardisation of managing CPI adverse events, providing consistency across the trusts.
Project Objectives
- To develop and standardise a regional immunotherapy education service for healthcare physicians across the Humber Health Partnership footprint
- Ensure that clinical staff are competent and confident in managing patients through their cancer journey when immunotherapy is used appropriately in suitable patients, across several differing tumour sites
- Improve service efficiency by reducing clinical time, moreover, reducing inpatient stays, or even allowing for patients to be managed in an outpatient setting.
Project Benefits
Patients
- A dedicated IO nurse led service may improve patients’ compliance with treatment schedules
- Patients will receive standardised and consistent education whilst on treatment with immunotherapy regardless of their cancer type
- Patients will have the ability to seek appropriate advice in a timely manner via the regional IO service, therefore has the potential to prevent unnecessary hospital attendance/admission
- Patients may report an improvement in the treatment end of their cancer pathway journey
NHS
- Patients managed by the IO CNS may result in releasing clinical capacity in other departments within the 2 trusts (e.g. accident & emergency, acute oncology services, primary care)
- Reduced length of inpatient stays in the acute oncology service wards due to patients and HCP being more confident at recognising onset of adverse events in a timelier manner.
- Efficient management of patient concerns will continually improve the cancer pathways of subsequent patients
- Data gathered from the project may be supportive, informative and compelling to lead the wider adoption of such initiatives in other trusts, therefore reducing variation of care within the NHS
MSD
- Reputational benefit of MSD working with the NHS to improve services
- A better understanding of both short- and long-term challenges associated with immunotherapy treatment options, and the impact on capacity within trusts to manage them successfully
- As a pharmaceutical manufacturer of oncology medicines, an indirect result could be that MSD may see use of their NICE approved medicines
Funding & Resources
This project is a shared contribution between Humber and North Yorkshire Cancer Alliance and MSD. The total project cost is £110,000
GB-NON-12046 | October 2025
Southern Health and Social Care Trusts Chemotherapy Day Unit Pathway Development Programme (PDP)
Project Title
Southern Health and Social Care Trusts Chemotherapy Day Unit Pathway Development Programme (PDP).
Organisations involved
Southern Health and Social Care Trust & MSD.
Summary
There is an opportunity in Southern Health and Social Care Trust to optimise the chemotherapy unit pathway to improve service quality, service efficiency, productivity and patient experience. The desired outcome of this project is to improve the efficiency of, or components of the chemotherapy unit pathway versus current baseline, including the achievement of the 31-day cancer waiting time targets for systemic anti-cancer therapy (SACT). The project is intended to run for approximately 12 months.
Background
The project is required to help address increasing pressure on the chemotherapy unit pathway, particularly in reducing waiting times and access to systemic anti-cancer therapy (SACT). Healthcare professionals, including consultants, nurses, and pharmacists, are currently overextended, with expectations of rising demand for services in the future.
Project Objectives
The project aims to establish a baseline understanding of the current timelines for each segment of the chemotherapy unit pathway through a comprehensive review involving multi-disciplinary teams. It seeks to identify inefficiencies that contribute to delays in patient transit along the pathway and produce a detailed map of the existing pathway along with a gap analysis. Southern Health and Social Care Trust may use the outputs to address identified inefficiencies, potentially improving outcomes for the chemotherapy unit patients and supporting the achievement of the 31-day cancer waiting time targets.
Benefits
Benefits to Patients
- An improved patient experience through the pan tumour chemotherapy day unit pathways in the Southern Health and Social Care Trust.
- Potentially quicker time to treatment (pan tumour) through improved pathway efficiencies.
Benefits to NHS
- Smoother and quicker referral process pan tumour following treatment decision.
- Potential improvement of the 31-day cancer waiting time target (pan tumour).
- Potential improvement in the model of delivery reducing capacity pressures.
- Potential efficiencies leading to more patients being treated faster.
Benefits to MSD
- Better understanding of the SACT patient journey.
- Enhanced reputation of MSD through partnership work.
- As a pharmaceutical manufacturer of oncology medicines, an indirect result of an improved pathway. may be that MSD see usage of their NICE/SMC reimbursed medicines.
Funding & Resources
This project is a shared contribution of time between Southern Health and Social Care Trust and MSD. No funding is associated with this collaborative working project.
GB-NON-12074 | October 2025
NHS Greater Glasgow & Clyde (GG&C) eMDT Project
Project Title
NHS Greater Glasgow & Clyde (GG&C) eMDT Project
Organisations involved
MSD and NHS Greater Glasgow & Clyde
Summary
NHS GG&C Multi-Disciplinary Team (MDT) have developed an electronic MDT (eMDT) platform which has been piloted and implemented in 2 regional tumour MDTs.
The purpose of this project is to support the eMDT team to scale the roll out of the platform to 15 cancer MDT’s (regional and local) in all Health Boards (HBs) in the West of Scotland Cancer Network (WoSCAN).
The project intends to run for 12 months.
Background
The NHS have developed an electronic MDT (eMDT) platform to deliver
- A common MDT solution for all cancers via a single, standardised platform,
- Tumour specific adaptations,
- Opportunities to improve patient pathways,
- Improved operational effectiveness for MDT’s,
- A standardised dataset to provide improved cancer intelligence,
- Business reporting functionality, and affordable solution
The eMDT team require additional resources and expertise to create a road map or implementation plan for the increased scale of work.
They require support with stakeholder mapping, influence mapping, project management and communication plans.
The team require additional resources or the expertise to scale the roll out to each tumour MDT in an efficient and timely way.
Project Objectives
- Complete stakeholder and influence mapping of local, regional and national stakeholders and creation of a stakeholder plan.
- Co-create a roadmap and implementation plan for roll-out to a new MDT/Health Board (HB).
- Co-create a communication plan including case studies.
- Develop an MDT HCP user survey to assess benefits realised with MDT improvement.
- Deliver the above for up to 5 eMDT tumour platforms in NHS GG&C during this one-year project. (Then handing over the blueprint plans for the NHS to take over the next 10 tumour groups).
- Fund a Band 7 FTE Launch Manager for 12 months only. The remit of this role would be to accelerate the delivery of the eMDT system across WoSCAN and transfer learning to other cancer networks in Scotland, supporting the MDT team with the transition between systems and educating users on the new platform. They would also provide training and benefits realisation for stakeholders, gather feedback and measure KPIs as well as problem solve any implementation issues here.
Benefits
Patients:
- As a result of streamlined MDT meetings, the patient may experience a faster cancer pathway (as measured by PHS Cancer wait time improvements).
- Improved care processes and optimal treatment decisions made for each patient.
NHS
- The benefits to patients and the NHS would mean that more MDT’s will be able to implement the tool more quickly and in a more efficient way.
- Improved MDT efficiency and quality of time spent discussing each patient leading to 62/31-day cancer waiting time improvements and smoother and quicker referral through the MDT and cancer pathway from the urgent suspicion of cancer referral to the decision to treat. Potentially efficiencies leading to more patients being treated in a faster time.
MSD
- Enhanced reputation of MSD through partnership work.
- A better understanding of MDT processes and outcomes.
- As a pharmaceutical manufacturer of oncology medicines, an indirect result of an improved. pathway may be that MSD see usage of their NICE / SMC approved medicines.
- A better understanding of NHS datasets relating the pathway and CWT’s.
Funding & Resources
This project is a shared contribution between NHS Greater Glasgow & Clyde and MSD.
GB-NON-11934 | October 2025
The Christie Ambulatory Service Expansion Project
Project Title
The Christie Ambulatory Service Expansion Project
Organisations involved
The Christie Cancer Centre NHS Foundation Trust & MSD
Summary
The Christie Ambulatory Service Expansion Project aims to address the increasing burden of treatment i.e. immunotherapy and chemotherapy, complications on hospital services. The project involves developing innovative ambulatory models for managing grade 3 and 4 complication, focusing on risk stratification and acute ambulatory management tools for selected patients in neoadjuvant and adjuvant settings.
The purpose of MSD providing funding to cover Advance Practitioner costs for a defined period of 18 months is to allow the Partner to produce a business case, after the conclusion of this project, to seek to have this funding continued and provided by the NHS on a permanent basis. MSD is therefore providing the Partner with limited funding needed to produce proof-of-concept, such that the NHS could assume responsibility for standing up an Advanced Practitioner (AP) on a permanent basis thereafter.
Project Objectives
- Enhance ambulatory management of immunotherapy complications for cancer patients.
- Reduce unnecessary inpatient admissions through effective risk stratification tools.
- Improve patient outcomes by providing prompt and comprehensive support.
- Develop a replicable model for other regions to improve patient experience and treatment continuity.
Deliverables
- Implementation of innovative ambulatory models.
- Development and deployment of risk stratification tools.
- To measure the success of the project, several key metrics will be closely monitored. These include tracking how many patients make use of the service, assessing how effectively complications are managed, evaluating the average duration of hospital stays, and analysing rates of patient satisfaction. Additionally, the rate at which patients disengage from the service will be reviewed, ensuring that any challenges or barriers to continued engagement can be identified and addressed.
- Creation of a regional best practice model for complication management.
Benefits
Patient Benefit:
- Improved management of treatment (immunotherapy and chemotherapy) complications. This will be calculated by tracking reductions in inpatient admissions and average length of stay for patients with grade 3 and 4 complications managed ambulatory. Additionally, monthly service utilisation data and patient satisfaction scores will be used to assess operational efficiency and care quality.
- Reduced inpatient bed days.
- Enhanced patient satisfaction and experience
Trust Benefit:
- Improved management of active treatment complications and release of capacity
- Sustained improvement of care, reduction of acute burden both in terms of attendance/admission and adverse outcomes/mortality
- Reduction in acute / unplanned admissions and related bed days
- Optimised patient management and continuity of treatment
MSD Benefit:
- Enhanced reputation for MSD through partnership work
- A better understanding of the challenges experienced along the treatment pathway to ensure informed conversations internally and with external stakeholders
Funding & Resources
This project is a shared contribution between The Christie Cancer Centre NHS Foundation Trust and MSD.
GB-NON-11864 | September 2025
West Yorkshire Non-Surgical Oncology (NSO) PDP
Project Title
West Yorkshire Non-Surgical Oncology (NSO) PDP
Organisations involved
Mid Yorkshire NHS Teaching Trust
Calderdale and Huddersfield NHS Foundation Trust
Summary
There is an opportunity in Calderdale and Huddersfield Foundation Trust (CHFT) and Mid Yorkshire NHS Teaching Trust (MYTT) to optimise the oral SACT pathways to improve service delivery and patient experience. The desired outcome of this project is to standardise SACT services across both trusts. The project intends to run for approximately 12 months.
Background
The Systemic Anti-Cancer Treatment (SACT) capacity and demand analysis conducted by MSD in 2024 for West Yorkshire and Harrogate Cancer Alliance highlighted that in one to two years’ time, the units at Calderdale and Huddersfield Foundation Trust (CHFT) and Mid Yorkshire NHS Teaching Trust (MYTT) will not have the chair capacity or qualified workforce for the demand.
To review the treatments delivered within the specialised units, and ensure care is close to the patient’s home where appropriate, a detailed mapping of the existing oral SACT pathways is now required. Current pathways across Calderdale and Huddersfield Foundation Trust (CHFT) and Mid Yorkshire NHS Teaching Trust (MYTT) vary, potentially affecting patient experience and the consistency of service delivery. Clear and coordinated pathway mapping is essential to standardise care, improving outcomes for patients as well as supporting new models of care and future demand.
Project Objectives
- Map existing oral SACT pathways for lung, prostate, and breast cancer within CHFT and MYTT.
- Identify opportunities to streamline patient flow, including exploring the feasibility of delivering oral and subcutaneous treatments outside of SACT chairs.
- Support pathway standardisation efforts to improve patient access, experience, and resource utilisation.
- Continue the achievement of 31-day and 62-day Cancer Waiting Time targets, ensuring earlier treatment days where possible.
Benefits
Benefits / Impact to Patients
- Improved patient experience of the NSO cancer pathway.
- Improved access to cancer care out of hospital-based SACT chairs.
- Reduced hospital visits, travel and time off work.
Benefits to the NHS Partner
- Optimisation of service delivery.
- Earlier treatment of lung, breast and prostate cancer patients improving the chance of successful treatment outcome.
- Increase in treatment rates for lung, breast, and prostate cancer.
- Achievement of the lung, breast and prostate 31-day treatment target and 62-day referral to treatment cancer waiting time targets.
Benefits to MSD
- Enhanced reputation of MSD through partnership work.
- As a pharmaceutical manufacturer of oncology medicines, an indirect result of an improved pathway may be that MSD see more appropriate usage of their NICE approved medicines.
Funding & Resources
This project is a shared contribution of time between Mid Yorkshire NHS Teaching Trust and Calderdale and Huddersfield NHS Foundation Trust and MSD.
GB-NON-11354 | May 2025
Re-design of a Specialist Immuno Oncology (SIO) Service supporting Systemic Anti-Cancer Treatment (SACT) delivery across Sussex
Project Title:
Re-design of a Specialist Immuno Oncology (SIO) Service supporting Systemic Anti-Cancer Treatment (SACT) delivery across Sussex
Organisations Involved
University Hospitals Sussex NHS Foundation Trust
East Sussex Healthcare NHS Trust
MSD UK
Summary
The project will support the development and delivery of a SIO Service providing different levels of support to patients and clinical teams across all six sites in East & West Sussex.
MSD (UK) were approached by a medical oncologist at UHSussex Brighton to provide project management support for the re-design of the existing acute oncology provision to develop a re-designed SIO Service. This is following a successful bid to Surrey & Sussex Cancer Alliance to fund the new/expanded specialist team for 24 months.
The new service will fill a gap in current provision by enabling equitable access for patients receiving immune checkpoint inhibitors (ICIs) immunotherapy to a highly skilled specialist team providing high quality Chemotherapy & Immunotherapy Supportive Care. The service will educate, provide advice & guidance to clinical teams around Sussex to identify toxicity early, helping to ensure treatment is effective and patients are well informed no matter where they are cared for.
The SIO team will predominately support patients experiencing side effects from immunotherapy cancer treatment but also implement management pathways which will include advice and guidance for other novel agents where a pathway can be optimised by the involvement of the service. The SIO team will additionally provide support and cross cover for their local trust SACT colleagues via a single ‘centralised’ team utilising digital processes and standard protocols.
The project will deliver the following as outcomes:
- Implementation and delivery of national management protocols, assessing /quantifying the movement of patients through the service (local pathways) and the development of new regional pathways
- Stratification of patient needs and provision of care and the interaction/transfer of care between these groups across the region, either under the SIO / Acute Oncology (AO) / Tumour Site Reference Group (TsRG) teams (including oncologists, site specific cancer nurse specialists, physician associates and specialist pharmacist)
- Develop equitable outpatient and ambulatory management, allowing admission avoidance in certain cases and expediting discharge for those for whom admission is necessary.
- Optimisation of management, triage and admission for patient with severe/ refractory adverse effects including the delivery of biological therapies.
- Appropriate utilisation, education & support of same day emergency care (SDEC) pathways.
- Development and delivery of patient facing resources to improve knowledge and patient experience to promote patient self-identification move towards self-management
MSD’s commitment will be for the agreed 24-month period. An evaluation will be completed at 30 months. The goal is to evaluate the impact on patient care, bed days and experience of the service. To achieve sustainability, 75% of the way through the 2-year program a joint case for investment will be developed and submitted UHSussex Exec +/- External Commissioners for substantive/permanent service funding beyond the initial transformation funding committed by SSCA (2024/2025).
Background
University Hospitals Sussex (UHSussex) NHS Foundation trust provides acute and specialist services for 6 hospitals across East and West Sussex including an oncology service in East Sussex Healthcare trusts serving a population of approx. 2.4 million people.
Delivery of SACT is recognised as a challenge at both regional and national levels. This is due to several issues including geographical disparity of pretreatment assessments, increased burden of assessment throughout treatment, sheer chair capacity, and the recognition, management, and support of patients during an acute problem with their cancer pathway.
Increasingly it is being recognised that there is a mortality burden associated with these newer therapeutics and avoidance of this may be optimised in the early recognition, appropriate management, and effective monitoring of patients.
Due to the burden of work on the Sussex cancer workforce including the increasing longevity of cancer care in palliative patient pathways, the delivery of direct patient care and education to the wider medical teams can be more effectively delivered by a specialised team supported by Standard Operating Procedures (SOPs) and decision matrices than the current piecemeal delivery by individual teams has resulted in inconsistencies of care and limited experiential learning up to this point.
Many cancer centres already provide nurse led clinic support at a tumour level or to manage the broader effects of immunotherapy e.g., Velindre Cancer Centre, Cardiff.
This project will aim to drive consistency of support across different cancers for patients treated with ICIs in all Trust led services across Sussex. The service will be important because it allows more patients to stay well whilst going through their cancer treatment and supports those who do get side effects so they are often less severe, with fewer negative impacts on the patient than there could be otherwise and to remain where possible on active therapy. Spotting the signs of these side effects early also means patients are less likely to be admitted to hospital.
Project Objectives
The objectives of this collaboration are as follows:
- Establishment of a dedicated service to run for 24 months within the scope of the project, followed by an evaluation 6 months after the project has completed. Within the scope of the project, a business case for ongoing trust support will be developed to sustain the service after the initial project period.
- Provide a specialist sub-acute management pathway to manage treatment continuance, wean treatment in a timely manner and co-ordinate team input where needed.
- Education and training for the TsRG teams to ensure robust, standardised care and continuity of medication management.
- Multi-speciality co-ordination and cross-speciality working to promote effective management of complex cases arising from improved depth of exposure and knowledge, underpinned by detailed patient record keeping.
Benefits
Benefits/ Impact to Patients
- Patients will receive consistent, standardised education on checkpoint inhibitors (CPIs) & how to manage the side effects during treatment irrespective of cancer type alongside their other tumour-related education.
- Patient will have a dedicated service and confidence that their immune treatment side effects are identified & managed appropriately in a timely manner
- Patients potentially stay on their treatment for longer/re-start their treatment following side effects
- Prevents unnecessary attendance / admission
Benefits to the NHS Partner
- Patients managed by the Specialist Immuno-Oncology (SIO) Service may result in released clinical capacity (oncology consultants/department CNS and Emergency Department (ED)/Acute medicine) that can be reallocated to the running of their clinics / other activity.
- Reduction in acute / unplanned admissions and related bed days
- Project management resource to support the development/re-design of the new SIO Service supporting SACT delivery across Sussex
- Sustained improvement of care, reduction of acute burden both in terms of attendance/admission and adverse outcomes/mortality
Benefits to MSD
- Enhanced reputation for MSD through partnership work
- A better understanding of the challenges experienced along the treatment pathway to ensure informed conversations internally and with external stakeholders
- As a pharmaceutical manufacturer of oncology medicines, and indirect result of the CPI is that MSD may see more appropriate use of their NICE approved medicines.
Funding & Resources
This project is a shared contribution of time between University Hospitals Sussex NHS Foundation Trust and MSD UK.
GB-NON-10761 | December 2024
Surrey & Sussex Cancer Alliance SACT Day Unit Demand & Capacity Project
Project Title
Surrey & Sussex Cancer Alliance SACT Day Unit Demand & Capacity Project
Organisations involved
Surrey & Sussex Cancer Alliance,
Ashford and St. Peter’s Hospitals NHS Foundation Trust,
University Hospitals Sussex NHS Foundation Trust,
East Sussex Healthcare NHS Trust,
Frimley Health NHS Foundation Trust,
Queen Victoria Hospital NHS Foundation Trust,
Royal Surrey NHS Foundation Trust,
Surrey and Sussex Healthcare NHS Trust (SASH)
MSD
Summary
This project is to be conducted in partnership with Surrey & Sussex Cancer Alliance, and will impact the following Trusts: Ashford and St. Peter’s Hospitals NHS Foundation Trust, University Hospitals Sussex NHS Foundation Trust, East Sussex Healthcare NHS Trust , Frimley Health NHS Foundation Trust, Queen Victoria Hospital NHS Foundation Trust, Royal Surrey NHS Foundation Trust.
The aim of this project is to provide data-led insights into demand and capacity of systemic anti-cancer treatment (SACT) delivery in current and future services for Surrey & Sussex Cancer Alliance. This analysis will be conducted using MSD’s capacity insights tool (CIT*) and aims to support optimisation of cancer treatment services to ensure timely access to optimal treatment. The insights from the model will highlight gaps and opportunities within the services’ ability to deliver current and future SACT. Facilitated capacity workshops (led by MSD) will enable a review of the insights report and a discussion on potential modelled scenarios that, if implemented by SACT day units, could address current/future service gaps. The data led decisions will be incorporated into an action plan for Trust cancer services to influence and implement changes. The intent is for SACT delivery service capacity to improve (measured via a second report from CIT) and therefore allow for more timely access to optimal treatments for Surrey & Sussex Cancer Alliance. The project is expected to run over a 12 month timeframe.
*CIT – The Capacity Insights Tool is a directional tool to support SACT delivery sites to understand how their capacity (in terms of chairs and workforce) can meet the patient demand on the service now and in the future. The tool can model future scenarios to illustrate how growing demand will impact the SACT delivery service and model the impact of potential changes. The report provided as a result of the tool can be used to identify which potential changes are needed to future proof the service.
Background
- SACT day units across the country are being challenged with increased demand stemming from increasing cancer patients, increasing SACT treatments, novel immunotherapy and combination treatments plus more treatments being delivered in the ambulatory setting. This increased demand for capacity plus workforce pressures are resulting in a stretched SACT delivery service sometimes resulting in delays to treatment for cancer patients.
- Many SACT managers have no, or limited access to tools to support their analysis of demand and capacity, have limited time and/or management processes to develop their demand and capacity data into actionable insights. This degrades their ability to address capacity issues due to their operational pressures.
- Cancer alliances have been directed in their 2024 NHSE Planning Pack to evaluate bi-annually their demand and capacity of SACT service provision across their footprint.
- To understand future SACT delivery options and models, it is important to understand the current demand and capacity and plan for sustainability.
Project Objectives
- Visibility of current and future capacity and demand (workforce and chair capacity) within SACT day units across Surrey & Sussex Cancer Alliance.
- Optimised treatment timings and utilisation of services within SACT day units across Surrey & Sussex Cancer Alliance.
- Improved cancer waiting times to begin SACT in line with national guidance
- Trust level service development initiatives implemented based on insights generated through this work
Benefits
Patient Benefits
- Reduced delay to treatment
- Reduction in ‘on the day’ delay and deferrals
NHS Benefits
- Increased treatment capacity to fulfil current and future demand
- Surrey & Sussex Cancer Alliance has an overview of their SACT services demand and capacity status to help deliver a more regional approach recommending a plan optimising resource use and highlighting where additional resource is required
MSD Benefits
- Enhanced reputation for MSD through partnership work
- Better understanding of treatment services and improvement opportunities
- As a pharmaceutical manufacturer of oncology medicines, an indirect result of improved treatment capacity may be that MSD see more appropriate usage of their NICE/SMC approved medicines
Funding & Resources
This project is a shared contribution of time between Surrey & Sussex Cancer Alliance and MSD.
GB-NON-10771 | December 2024
SACT Capacity and Demand Modelling Service
Project Title
SACT Capacity and Demand Modelling Service
Organisations Involved
Northern Cancer Alliance
Newcastle Hospitals NHS Foundation Trust
South Tees Hospital NHS Foundation Trust
County Durham and Darlington NHS Foundation Trust
North Cumbria Integrated Care NHS Foundation Trust
Northumbria Healthcare NHS Trust
South Tyneside and Sunderland NHS Foundation Trust
Gateshead Health NHS Foundation Trust
North Tees and Hartlepool NHS Foundation Trust
MSD UK
Summary
This project is to be conducted in partnership with Northern Cancer Alliance and will impact the following Trusts: Newcastle Hospitals NHS Foundation Trust, South Tees Hospital NHS Foundation Trust, County Durham and Darlington NHS Foundation Trust, North Cumbria Integrated Care NHS Foundation Trust, Northumbria Healthcare NHS Trust, South Tyneside and Sunderland NHS Foundation Trust, Gateshead Health NHS Foundation Trust, North Tees and Hartlepool NHS Foundation Trust.
The aim of this project is to provide data-led insights into demand and capacity of systemic anti-cancer treatment (SACT) delivery in current and future services for Northern Cancer Alliance. This analysis will be conducted using MSD’s capacity insights tool (CIT*) and aims to support optimisation of cancer treatment services to ensure timely access to optimal treatment. The insights from the model will highlight gaps and opportunities within the services’ ability to deliver current and future SACT. Facilitated capacity workshops (led by MSD) will enable a review of the insights report and a discussion on potential modelled scenarios that, if implemented by SACT day units, could address current/future service gaps. The data led decisions will be incorporated into an action plan for Trust cancer services to influence and implement changes. The intent is for SACT delivery service capacity to improve (measured via a second report from CIT) and therefore allow for more timely access to optimal treatments for Northern Cancer Alliance. The project is expected to run over a 12-month timeframe.
*CIT – The Capacity Insights Tool is a directional tool to support SACT delivery sites to understand how their capacity (in terms of chairs and workforce) can meet the patient demand on the service now and in the future. The tool can model future scenarios to illustrate how growing demand will impact the SACT delivery service and model the impact of potential changes. The report provided as a result of the tool can be used to identify which potential changes are needed to future proof the service.
Background
- SACT day units across the country are being challenged with increased demand stemming from increasing cancer patients, increasing SACT treatments, novel immunotherapy and combination treatments plus more treatments being delivered in the ambulatory setting. This increased demand for capacity plus workforce pressures are resulting in a stretched SACT delivery service sometimes resulting in delays to treatment for cancer patients.
- Many SACT managers have no, or limited access to tools to support their analysis of demand and capacity, have limited time and/or management processes to develop their demand and capacity data into actionable insights. This degrades their ability to address capacity issues due to their operational pressures.
- Cancer alliances have been directed in their 2024 NHSE Planning Pack to evaluate bi-annually their demand and capacity of SACT service provision across their footprint.
- To understand future SACT delivery options and models, it is important to understand the current demand and capacity and plan for sustainability.
Project Objectives
- Visibility of current and future capacity and demand (workforce and chair capacity) within SACT day units across Northern Cancer Alliance
- Optimised treatment timings and utilisation of services within SACT day units across Northern Cancer Alliance
- Improved cancer waiting times to begin SACT in line with national guidance
- Trust level service development initiatives implemented based on insights generated through this work
Benefits
Patient Benefits
- Reduced delay to treatment
- Reduction in ‘on the day’ delay and deferrals
NHS Benefits
- Increased treatment capacity to fulfil current and future demand
- Northern Cancer Alliance has an overview of their SACT services demand and capacity status to help deliver a more regional approach recommending a plan optimising resource use and highlighting where additional resource is required
MSD Benefits
- Enhanced reputation for MSD through partnership work
- Better understanding of treatment services and improvement opportunities
- As a pharmaceutical manufacturer of oncology medicines, an indirect result of improved treatment capacity may be that MSD see more appropriate usage of their NICE/SMC approved medicines
Funding & Resources
This project is a shared contribution of time between Northern Cancer Alliance and MSD.
GB-NON-10352 | November 2024
SACT Capacity and Demand Modelling service, West Yorkshire & Harrogate Cancer Alliance
Project Title
SACT Capacity and Demand Modelling service, West Yorkshire & Harrogate Cancer Alliance
Organisations involved
West Yorkshire & Harrogate Cancer Alliance
Leeds Teaching Hospital NHS Trust
Mid Yorkshire Teaching Hospitals NHS Trust
Harrogate and District NHS Foundation Trust
Calderdale and Huddersfield NHS Foundation Trust
Bradford Teaching Hospitals NHS Trust
Airdale NHS Foundation Trust
MSDUK
Summary
This project is to be conducted in partnership with West Yorkshire and Harrogate Cancer Alliance and will impact the following Trusts: Leeds Teaching Hospital NHS Trust, Mid Yorkshire Teaching Hospitals NHS Trust, Harrogate and District NHS Foundation Trust, Calderdale and Huddersfield NHS Foundation Trust, Bradford Teaching Hospitals NHS Trust, Airedale NHS Foundation Trust.
The aim of this project is to provide data-led insights into demand and capacity of systemic anti-cancer treatment (SACT) delivery in current and future services for West Yorkshire and Harrogate Cancer Alliance. This analysis will be conducted using MSD’s capacity insights tool (CIT*) and aims to support optimisation of cancer treatment services to ensure timely access to optimal treatment. The insights from the model will highlight gaps and opportunities within the services’ ability to deliver current and future SACT. Facilitated capacity workshops (led by MSD) will enable a review of the insights report and a discussion on potential modelled scenarios that, if implemented by SACT day units, could address current/future service gaps. The data led decisions will be incorporated into an action plan for Trust cancer services to influence and implement changes. The intent is for SACT delivery service capacity to improve (measured via a second report from CIT) and therefore allow for more timely access to optimal treatments for West Yorkshire and Harrogate Cancer Alliance The project is expected to run over a 12 month timeframe.
*CIT – The Capacity Insights Tool is a directional tool to support SACT delivery sites to understand how their capacity (in terms of chairs and workforce) can meet the patient demand on the service now and in the future. The tool can model future scenarios to illustrate how growing demand will impact the SACT delivery service and model the impact of potential changes. The report provided as a result of the tool can be used to identify which potential changes are needed to future proof the service.
Background
- SACT day units across the country are being challenged with increased demand stemming from increasing cancer patients, increasing SACT treatments, novel immunotherapy and combination treatments plus more treatments being delivered in the ambulatory setting. This increased demand for capacity plus workforce pressures are resulting in a stretched SACT delivery service sometimes resulting in delays to treatment for cancer patients
- Many SACT managers have no, or limited access to tools to support their analysis of demand and capacity, have limited time and/or management processes to develop their demand and capacity data into actionable insights. This degrades their ability to address capacity issues due to their operational pressures
- Cancer alliances have been directed in their 2024 NHSE Planning Pack to evaluate bi-annually their demand and capacity of SACT service provision across their footprint
- To understand future SACT delivery options and models, it is important to understand the current demand and capacity and plan for sustainability
Project Objectives
- Visibility of current and future capacity and demand (workforce and chair capacity) within SACT day units across West Yorkshire and Harrogate Cancer Alliance
- Optimised treatment timings and utilisation of services within SACT day units across West Yorkshire and Harrogate Cancer Alliance
- Improved cancer waiting times to begin SACT in line with national guidance
- Trust level service development initiatives implemented based on insights generated through this work
Benefits
Patient Benefits
- Reduced delay to treatment
- Reduction in ‘on the day’ delay and deferrals
NHS Benefits
- Increased treatment capacity to fulfil current and future demand
- West Yorkshire and Harrogate Cancer Alliance has an overview of their SACT services demand and capacity status to help deliver a more regional approach recommending a plan optimising resource use and highlighting where additional resource is required
MSD Benefits
- Enhanced reputation for MSD through partnership work
- Better understanding of treatment services and improvement opportunities
- As a pharmaceutical manufacturer of oncology medicines, an indirect result of improved treatment capacity may be that MSD see more appropriate usage of their NICE/SMC approved medicines
Funding & Resources
This project is a shared contribution of time between West Yorkshire and Harrogate Cancer Alliance and MSD.
GB-NON-10230 | September 2024
South Eastern Health and Social Care Trust Chemotherapy Day Unit Pathway Development Programme (PDP)
Project Title
South Eastern Health and Social Care Trust Chemotherapy Day Unit Pathway Development Programme (PDP)
Organisations involved
MSD
South Eastern Health and Social Care Trust
Summary
There is an opportunity in South Eastern Health and Social Care Trust to optimise the Chemotherapy Day Unit pathway to improve the understanding of the current patient pathway for each tumour site served, understand the challenges of these pathways and identify ways to address these challenges. It will also look at a consistent referral approach defined and implemented across all specialty’s to potentially improve treatment time and MDT decision making to progress patients through the treatment pathway in the Chemotherapy unit. The project intends to run for approximately 12 months.
Project Objectives
The primary objective of this project is the optimisation of the Chemotherapy Day Unit Pan Tumour pathways across South Eastern Health and Social Care Trust. Specifically contributing towards; –
- Improving the 31-day treatment target Cancer Waiting Time targets
Benefits
Patient Benefits
- An improved patient experience through pan tumour Chemotherapy Unit pathways in the South Eastern Health and Social Care Trust
- Quicker time to treatment pan tumour, improving the chance of successful treatment
NHS Benefits
- Smoother & Quicker Referral Process pan tumour following treatment decision
- Improvement of the Pan Tumour 31-day Cancer Waiting Time targets
- Optimisation of service delivery through the Chemotherapy Day Unit
MSD Benefits
- Better understanding of chemotherapy patient journey
- Enhanced reputation of MSD through partnership work
- As a pharmaceutical manufacturer of oncology medicines, an indirect result of an improved pathway may be that MSD see more appropriate usage of their NICE/SMC approved medicines
Funding
No funding is associated with this project. The project is a shared contribution of time from both the NHS organisation and MSD
GB-NON-09926 | September 2024
SACT Capacity and Demand
Project Title
SACT Capacity and Demand
Organisations involved
South Yorkshire and Bassetlaw Cancer Alliance
Sheffield Teaching Hospitals NHS foundation Trust
Barnsley Hospital NHS Foundation Trust
Rotherham NHS Foundation Trust
Doncaster and Bassetlaw Teaching Hospital NHS Foundation Trust
Chesterfield Royal Hospital NHS Foundation Trust
Summary
The aim of this project over a 12-month period is to provide data-led insights into demand and capacity of systemic anti-cancer treatment (SACT) delivery in current and future services for South Yorkshire & Bassetlaw Cancer Alliance. This analysis will be conducted using MSD’s capacity insights tool (CIT*) and aims to support optimisation of cancer treatment services to ensure timely access to optimal treatment. The insights from the model will highlight gaps and opportunities within the services’ ability to deliver current and future SACT. Facilitated capacity workshops (led by MSD) will enable a review of the insights report and a discussion on potential modelled scenarios that, if implemented by SACT day units, could address current/future service gaps. The data led decisions will be incorporated into an action plan for Trust cancer services to influence and implement changes. The intent is for SACT delivery service capacity to improve (measured via a second report from CIT) and therefore allow for more timely access to optimal treatments for South Yorkshire & Bassetlaw Cancer Alliance.
Background
SACT day units across the country are being challenged with increased demand stemming from increasing cancer patients, increasing SACT treatments, novel immunotherapy and combination treatments plus more treatments being delivered in the ambulatory setting. This increased demand for capacity, plus workforce pressures, are resulting in a stretched SACT delivery service sometimes resulting in delays to treatment for cancer patients.
Many SACT managers have no, or limited access to tools to support their analysis of demand and capacity, have limited time and/or management processes to develop their demand and capacity data into actionable insights. This degrades their ability to address capacity issues due to their operational pressures.
Cancer Alliances have been directed in their 2024 NHSE Planning Pack to evaluate bi-annually their demand and capacity of SACT service provision across their footprint.
To understand future SACT delivery options and models, it is important to understand the current demand and capacity and plan for sustainability.
Project Objectives
To deliver two rounds of capacity insights and scenario planning delivered via PDF reports for all SACT day units within the project.
Identification of potential improvement activities through a capacity planning workshop and a corresponding action plan for SACT day units to implement to address gaps in current and future SACT delivery service capacity.
Evaluation report of the impact of changes made to demonstrate whether the SACT delivery service capacity across South Yorkshire & Bassetlaw Cancer Alliance has been optimised because of the project.
Benefits
Patients
Reduces delay to SACT treatment
Reduction in the ‘on the day’ delays and deferrals
NHS
Increased treatment capacity to fulfil current and future demand.
An oversight of their SACT service demand and capacity status to help deliver a more regional approach recommending a plan optimising resource use and highlighting where additional resource is required.
MSD
Enhanced reputation for MSD through partnership work
Better understanding of SACT treatment services and improvement opportunities
As a pharmaceutical manufacturer of oncology medicines, an indirect result of improved capacity may be that MSD see more appropriate usage of NICE/SMC approved medicines
Funding & Resources
This project is a shared contribution of time between South Yorkshire and Bassetlaw Cancer Alliance, Sheffield Teaching Hospitals NHS foundation Trust. Barnsley Hospital NHS Foundation Trust, Rotherham NHS Foundation Trust, Doncaster and Bassetlaw Teaching Hospital NHS Foundation Trust, Chesterfield Royal Hospital NHS Foundation Trust and MSD.
GB-NON-09875 | August 2024
Kent & Medway Cancer Alliance SACT Day Unit Demand & Capacity Project
Project Title
Kent & Medway Cancer Alliance SACT Day Unit Demand & Capacity Project
Organisations involved
Kent & Medway Cancer Alliance (Kent & Medway Integrated Care Board (ICB)), MSD, Dartford & Gravesham NHS Trust, East Kent University hospital NHS Trust, Maidstone and Tunbridge wells NHS Trust, Medway Foundation NHS Trust
Summary
This project is to be conducted in partnership with Kent & Medway Cancer Alliance and will impact the following Trusts: Dartford & Gravesham NHS Trust, East Kent University hospital NHS Trust, Maidstone and Tunbridge wells NHS Trust, Medway Foundation NHS Trust The aim of this project is to provide data-led insights into demand and capacity of systemic anti-cancer treatment (SACT) delivery in current and future services for Kent & Medway Cancer Alliance This analysis will be conducted using MSD’s capacity insights tool (CIT*) and aims to support optimisation of cancer treatment services to ensure timely access to optimal treatment. The insights from the model will highlight gaps and opportunities within the services’ ability to deliver current and future SACT. Facilitated capacity workshops (led by MSD) will enable a review of the insights report and a discussion on potential modelled scenarios that, if implemented by SACT day units, could address current/future service gaps. The data led decisions will be incorporated into an action plan for Trust cancer services to influence and implement changes. The intent is for SACT delivery service capacity to improve (measured via a second report from CIT) and therefore allow for more timely access to optimal treatments for Kent & Medway Cancer Alliance The project is expected to run over a 12 months timeframe.
*CIT – The Capacity Insights Tool is a directional tool to support SACT delivery sites to understand how their capacity (in terms of chairs and workforce) can meet the patient demand on the service now and in the future. The tool can model future scenarios to illustrate how growing demand will impact the SACT delivery service and model the impact of potential changes. The report provided as a result of the tool can be used to identify which potential changes are needed to future proof the service.
Background
- SACT day units across the country are being challenged with increased demand stemming from increasing cancer patients, increasing SACT treatments, novel immunotherapy and combination treatments plus more treatments being delivered in the ambulatory setting. This increased demand for capacity plus workforce pressures are resulting in a stretched SACT delivery service sometimes resulting in delays to treatment for cancer patients.
- Many SACT managers have no, or limited access to tools to support their analysis of demand and capacity, have limited time and/or management processes to develop their demand and capacity data into actionable insights. This degrades their ability to address capacity issues due to their operational pressures.
- Cancer alliances have been directed in their 2024 NHSE Planning Pack to evaluate bi-annually their demand and capacity of SACT service provision across their footprint.
- To understand future SACT delivery options and models, it is important to understand the current demand and capacity and plan for sustainability.
Project Objectives
- Visibility of current and future capacity and demand (workforce and chair capacity) within SACT day units across Kent & Medway Cancer Alliance.
- Optimised treatment timings and utilisation of services within SACT day units across Kent & Medway Cancer Alliance.
- Improved cancer waiting times to begin SACT in line with national guidance
- Trust level service development initiatives implemented based on insights generated through this work
Benefits
Patient Benefits
- Reduced delay to treatment
- Reduction in ‘on the day’ delay and deferrals
NHS Benefits
- Increased treatment capacity to fulfil current and future demand
- Kent & Medway Cancer Alliance has an overview of their SACT services demand and capacity status to help deliver a more regional approach recommending a plan optimising resource use and highlighting where additional resource is required
MSD Benefits
- Enhanced reputation for MSD through partnership work
- Better understanding of treatment services and improvement opportunities
- As a pharmaceutical manufacturer of oncology medicines, an indirect result of improved treatment capacity may be that MSD see more appropriate usage of their NICE/SMC approved medicines
Funding & Resources
This project is a shared contribution of time between Kent & Medway Cancer Alliance and MSD.
GB-NON-09806 | August 2024
Western Health and Social Care Trusts Chemotherapy Day Unit Pathway Development Programme (PDP)
Project Title
Western Health and Social Care Trusts Chemotherapy Day Unit Pathway Development Programme (PDP)
Organisations involved
MSD
Western Health and Social care trusts
Summary
There is an opportunity in Western Health and Social Care trusts to optimise the Chemotherapy Day Unit pathway to improve the service quality, service efficiency, productivity and patient experience. The desired outcome of this project is improvement of the Chemotherapy Day Unit patient pathway and of the Pan Tumour 31-day Cancer Waiting Time Target. The project intends to run for approximately 12 months.
Project Objectives
The primary objective of this project is the optimisation of the Chemotherapy Day Unit Pan Tumour pathways across Western Health and Social Care Trust. Specifically contributing towards; –
- Improving the 31-day treatment target Cancer Waiting Time targets
- Measurement of the outcomes of the project and disseminating these outcomes within 6 months of the project completion
Benefits
Patient Benefits
- An improved patient experience of the Chemotherapy Day Unit pathways in the Western Health and Social Care Trust
- Faster time to treatment pan tumour and hence improving treatment options
NHS Benefits
Optimised Pan Tumour pathways in the Chemotherapy Day Unit across Western Health and Social Care trust hospital sites resulting in
- Smoother and Quicker time to treatment process following treatment decision
- Potential to improve the pan tumour 31-day cancer waiting time target
- Optimisation of service delivery through the Chemotherapy Day Unit
MSD Benefits
- Better understanding of chemotherapy patient journey through the Chemotherapy Day Unit and patients requiring hospital admission
- Enhanced reputation of MSD through partnership work
- As a pharmaceutical manufacturer of oncology medicines, an indirect result of an improved pathway may be that MSD see more appropriate usage of their NICE/SMC approved medicines
Funding
No funding is associated with this project. The project is a shared contribution of time from both the NHS organisation and MSD
GB-NON-09634 | July 2024
South Yorkshire & Bassetlaw Cancer Alliance MDT optimisation project for Lung, Skin, Urology and Upper gastrointestinal (UGI).
Project Title
South Yorkshire & Bassetlaw Cancer Alliance MDT optimisation project for Lung, Skin, Urology and Upper gastrointestinal (UGI).
Organisations involved
- South Yorkshire & Bassetlaw Cancer Alliance
- Sheffield Teaching Hospitals Foundation trust
- Doncaster & Bassetlaw Foundation trust
- Rotherham Foundation Trust
- Barnsley Foundation Trust
- Chesterfield Foundation Trust
- MSD UK (Ltd)
Summary
The project will provide project management support 1.5 days per week for 12 months to support South Yorkshire and Bassetlaw Cancer Alliance to implement MDT optimisation in Lung, Skin, Urology and Upper Gastrointestinal (UGI) tumour sites in the 5 foundation trusts – Sheffield, Doncaster & Bassetlaw, Rotherham, Barnsley, and Chesterfield.
The pre-MDT standards of care will be developed by the Trusts in conjunction with the Cancer Alliance, and MSD will support in implementing the standards across the local and regional MDTs. MSD will only be involved in the implementation and not the development of the standards. The project will aim to drive forward National recommendations and guidance for MDTs to deliver an improved MDT process (1). This will be done by ensuring streamlined processes/ standards of care pathways are developed and implemented to make the best use of clinical time and resources therefore improving the efficiency of the MDT meetings.
As a result, patients may benefit from an accelerated review through the pathway, either in MDT, or outside of MDT, using pre-agreed protocols for those less urgent or severe cases. This could allow for treatment decision to be made earlier and therefore earlier access to treatment, moreover reducing patient anxiety and potentially improved access to personalised treatment options, and will also support the NHS trusts to achieve the NHSE 62 day cancer wait time target.
Background
Research has shown that by 2035, the number of patients diagnosed with cancer in the UK could reach 500,000 (1), a material increase from the 357,000 who were diagnosed in 2014.
Multidisciplinary Team meetings are seen as the ‘gold standard’ to cancer management whereby a cross sector of health professionals including of histopathologists, radiologists, surgeons, cancer nurse specialists, oncologists, and administrators/MDT coordinators with additional members dependent on the individual cancer tumour site meet to discuss individual patients’ cases and make treatment recommendations. Multi-disciplinary working continues throughout the patient pathway through diagnosis to treatment and ongoing care however the multi-disciplinary team meeting is a key discussion forum where cases are presented, a range of information shared and reviewed, and care management discussed and documented.
MDT meetings typically occur weekly and can last several hours. (2) Staff increases have not kept pace with the increase in the number of cases and so this has meant that proportionately more of clinician’s time is spent preparing for and being involved in MDT discussions with a significant amount of organisation, scheduling, clinical input and administration time/oversight processes are involved in MDT working in general and more specifically in preparing/contributing to and following up to/from the MDT-meeting. The number of patient discussions per MDT has increased with proportionately the amount of time available for each patient discussion being reduced.
Project Objectives
- An assessment of MDT meetings operating efficiencies within lung, skin, Urology, upper GI pathways across the South Yorkshire & Bassetlaw region.
- Identification of improvement initiatives to address inefficiencies linked to MDT streamlining principles and promote geographical equitable patient experience of care across all the foundation trusts.
- Provide a regional consistent approach of MDT referrals via proformas with minimum MDT referral data sets for both trust level MDTs, and regional specialist MDTs to drive quicker treatment decisions.
- Development and Implementation of localised standards of care which will be based on national or international standards, guidelines and protocols, and best practice as determined by the Cancer Alliance tumor pathway board.
Benefits
Patient
- A potential for earlier communication of a treatment plan, due to a more efficient MDT process.
- Through reduced delays, more efficient communication to patient from the MDT, and therefore the potential for less anxiety related to process.
- Improved patient outcomes.
NHS
- Improved cancer wait times to support achievement of NHS 28 -day and 62-day cancer wait time targets.
- Standardisation and optimisation of practice through sharing of knowledge and experience.
- Fiscal savings due to decrease in patient breaches.
MSD
- Greater understanding of MDT process and decision making within the NHS.
- Increased collaborative working opportunities through building trust and effecting change.
- The intended collaboration may enable more patients to have access to treatment options in line with NICE guidelines which may or may not include MSD medicines.
Funding & Resources
This project is a shared contribution of time between South Yorkshire & Bassetlaw Cancer Alliance and MSD.
GB-NON-13146 | June 2026
(1)Cancer Research UK (2017) Meeting Patients Needs – Improving the Effectiveness of Multidisciplinary Team Meetings in Cancer Services, https://www.cancerresearchuk.org/sites/default/files/full_report_meeting_patients_needs_improving_the_effectiveness_of_multidisciplinary_team_meetings_.pdf [Accessed May 2024]
(2) Independent Cancer Taskforce. (2015). Achieving World-Class Cancer Outcomes: A Strategy for England 20152020. London: Independent Cancer Taskforce. https://www.cancerresearchuk.org/sites/default/files/achieving_world-class_cancer_outcomes_-_a_strategy_for_england_2015-2020.pdf [Accessed May 2024]
Pan Tumour – Completed Projects
Patient Education Programme – The Clatterbridge Cancer Centre NHS Foundation Trust
Project Title
Patient Education Programme – The Clatterbridge Cancer Centre NHS Foundation Trust
Organisations involved
MSD UK Ltd and The Clatterbridge Cancer Centre NHS Foundation Trust
Summary
The objectives of this collaboration are as follows: The proposed pilot was delivered as a collaboration between MSD and The Clatterbridge Cancer Centre. MSD provided funding, along with project management support, to support the recruitment of a Patient Education Programme Lead. This role was funded by MSD for a 12-month period only. The Programme Lead at Clatterbridge Cancer Centre developed patient education workshops (breast/skin/urology) using existing materials, trained relevant team members to deliver the workshops, and monitored and audited implementation of workshops. They collated patient resources and clinician training materials to support the patient across the treatment pathway. The patient resources related to cancer and information on what to expect throughout the patient journey and did not relate to any individual drug.
The aim was to provide consistency, monitoring and review of the Patient Education Programme and to ensure all resources remained up to date and regularly audited.
The project successfully delivered a structured patient education offer, including tumour-specific workshops, digital resources and tools to support self-management. A key enabler was the implementation of the Patients Know Best (PKB) portal, providing patients with access to information, recorded content and digital tools to support their care outside of clinical settings.
Overall, the main deliverables were achieved. However, progress varied across tumour groups, and some elements require further time to embed fully. In particular, consistent rollout across all hubs, workforce training for delivery, and routine monitoring of uptake and experience are still being developed.
Benefits Realised
Benefits to Patients
- Patients now have improved access to clear, structured education resources covering diagnosis, treatment, side effects and follow-up care.
- Digital access via Patients Know Best (PKB) portal has enabled patients to engage with education materials at a time and place that suits them, including recorded workshops and personalised resources.
- Remote follow-up pathways and tools (e.g. electronic Patient-Reported Outcome Measures (ePROMs), symptom trackers) have supported greater patient involvement in self-management and monitoring of their condition.
- Patient feedback indicates that digital pathways are easy to use, provide reassurance, and help maintain a sense of connection with clinical teams.
The programme has helped address key unmet needs, including emotional support, symptom management, and clearer follow-up communication.
Benefits to the NHS
- A structured and standardised approach to patient education has been established across multiple tumour pathways, improving consistency of information provided to patients.
- Digital infrastructure via the PKB portal has enabled scalable access to education resources, supporting patients beyond traditional clinic settings.
- New digital pathways, including remote follow-up models and education-integrated care pathways, have been developed to support personalised patient management.
- Workshops and education resources have been developed, piloted, and refined using patient and clinician feedback, improving the quality and relevance of information delivered.
The programme has supported improved communication, signposting and access to wider support services such as psychological, financial and lifestyle support.
Benefits to MSD
- Strengthened collaborative working with The Clatterbridge Cancer Centre, supporting shared goals to improve services and patient experience.
This collaboration may have enabled more patients to access innovative treatments in line with NICE guidance which may or may not have included MSD medicines.
Programme Outcomes
Implementation and Uptake
Over a 12-month period, a total of 158 patient education workshops were delivered and accessed by patients. Engagement was observed across multiple service areas, including:
- Breast: 350 patient attendances
- Urology: 189 patient attendances
- Skin: 65 patient attendances
These figures describe activity levels associated with the implementation of the education programme.
Use of Existing Patient Resources
Engagement with patient education materials hosted on the Clatterbridge intranet was monitored. Recorded activity included:
- Breast: 1,714 patient clicks
- Urology: 1,602 patient clicks
- Skin: Evaluation ongoing
This data reflects utilisation of existing digital resources made available to patients.
Healthcare Professional Feedback
Feedback from healthcare professionals (HCPs) was collected through qualitative questionnaires exploring their experience of the education resources and workshops.
Responses indicated that the resources and workshops were considered acceptable for use in practice. Feedback also identified a preference for a combination of digital and face-to-face delivery approaches to support patient education.
Patient Feedback
Patient experience was assessed through qualitative feedback and observation of engagement patterns.
- Initial feedback indicated that patients found the resources accessible and easy to use
- Increased website activity was observed during the evaluation period
- Further formal evaluation is planned following the embedding of digital access pathways
Illustrative feedback from patients participating in the breast workshop pilot and PKB (Patients Know Best) included:
- “PKB was easy to navigate”
- “Remote follow-up felt simple and clear”
Important Information
All patient education materials referenced were pre-existing resources; no new promotional or educational materials were created as part of this collaboration
The content of the resources was non-promotional and related to general cancer information and patient support throughout the treatment pathway
No information relating to specific medicines was included
Funding & Resources
This project was a shared funding commitment from The Clatterbridge Cancer Centre & MSD. The total project cost was £51,444
Lessons learnt
- Baseline patient and staff feedback is critical to identifying meaningful gaps and prioritising education improvements.
- A blended delivery model (face-to-face and digital) is essential to ensure equitable access, given workforce constraints and varying patient preferences.
- Digital platforms such as PKB can support standardisation and scalability, but successful implementation depends on clinical pathway readiness, digital onboarding and operational capacity.
- Patients value clear, simple and practical information, particularly following treatment when ongoing clinical contact is reduced.
- Robust data collection and reporting mechanisms are required to demonstrate impact and support continuous improvement.
- Close collaboration and regular touchpoints between NHS project leadership and the MSD Project Manager played an important role in driving delivery and supporting the continued sustainability of the programme.
Publications
There are no planned external publications or congress presentations associated with this project.
Reference
- The Clatterbridge Cancer Centre Patient Education Project report
GB-NON-13045 | May 2026
Royal Marsden Partners Cancer Alliance (RMPCA) SACT Capacity and Demand Project
Project Title
Royal Marsden Partners Cancer Alliance (RMPCA) SACT Capacity and Demand Project
Organisations involved
- RM Partners
- MSD
- Croydon Health Services NHS Trust
- Chelsea and Westminster Hospital NHS Foundation Trust
- Epsom and St Helier University Hospitals NHS Trust
- Kingston Hospital NHS Foundation Trust
- London Northwest University Healthcare NHS Trust
- Imperial College Healthcare NHS Trust
- St George’s University Hospitals NHS Foundation Trust
- The Hillingdon Hospitals NHS Foundations Trust
- The Royal Marsden NHS Foundation Trust
Summary
The outcome summary for the RMPCA Systemic Anti‑Cancer Therapy (SACT) Capacity and Demand Project is currently pending due to finalisation and alignment between parties. We remain committed to publishing the full results once the required information is received and validated.
This 12-month collaborative working project was delivered in partnership with Royal Marsden Partners and NHS Trusts across the region (listed above). The project focused on developing a robust, data‑led understanding of SACT demand and capacity, to support service optimisation and sustainability across RMPCA.
The objectives of this collaboration were as follows:
- Visibility of current and future capacity and demand (workforce and chair capacity) within SACT delivery sites across the cancer alliance.
- Optimised treatment and utilisation of services across at SACT delivery sites.
- Improved patient experience during SACT delivery across SACT delivery sites.
- Improve cancer waiting times to begin SACT in line with national guidance.
- Standardisation of practices in SACT delivery.
Using MSD’s Capacity Insights Tool (CIT), data was gathered from NHS SACT units and used to model current and future service needs, identifying gaps and opportunities to optimise delivery. Reports and insights were shared and reviewed at facilitated workshops and scenarios captured to improve patient access and efficiencies were explored.
This project supported NHS England’s (NHSE) 2024/2025 directive for biannual SACT capacity reviews and responds to rising demand, complex treatments, and workforce pressures. Outcomes include reduced delay to treatment, improved experience of treatment due to improved capacity and reduction in ‘on the day’ delay and deferrals.
Next Steps
- Partner organisations will align on the data and associated outcomes.
- Once agreed, the outcome summary will be finalised and submitted for certification.
- We will update stakeholders promptly as progress is made.
Note:
This holding statement ensures transparency and compliance while we work collaboratively to complete the project deliverables.
Benefits Realised
Data and outcomes are currently being reviewed and will be updated in due course.
Publications
No publications are planned outside of this holding statement.
GB-NON-13082 | May 2026
North East London Cancer Alliance SACT Capacity and Demand Project
Project Title
North East London Cancer Alliance SACT Capacity and Demand Project
Organisations involved
- North East London Cancer Alliance c/o North East London ICB
- Barts Health NHS Trust
- Barking, Havering and Redbridge University Hospitals NHS Trust
- MSD
Summary
The outcome summary for the North East London Cancer Alliance SACT Capacity and Demand Project is currently pending due to NHS restructure currently underway delaying the finalisation for the NHS partners. We remain committed to publishing the finalised outcome summary once NHS partners approval is provided.
This project is to be conducted in partnership with North East London Cancer Alliance and will impact the following Trusts: Barts Health NHS Trust & Barking, Havering and Redbridge University Hospitals NHS Trust.
The aim of this project is to provide data-led insights into demand and capacity of systemic anti-cancer treatment (SACT) delivery in current and future services for North East London Cancer Alliance. This analysis will be conducted using MSD’s capacity insights tool (CIT) and aims to support optimisation of cancer treatment services to ensure timely access to optimal treatment. The insights from the model will highlight gaps and opportunities within the services’ ability to deliver current and future SACT. Facilitated capacity workshops (led by MSD) will enable a review of the insights report and a discussion on potential modelled scenarios that, if implemented by SACT day units, could address current/future service gaps. The data led decisions will be incorporated into an action plan for Trust cancer services to influence and implement changes. The intent is for SACT delivery service capacity to improve (measured via a second report from CIT) and therefore allow for more timely access to optimal treatments for North East London Cancer Alliance. The project is expected to run over a 12 month timeframe with a start date of November 2024.
Next Steps:
- Partner organisations will align on the data and associated outcomes.
- Once agreed, the outcome summary will be finalised and submitted for certification.
- We will update stakeholders promptly as progress is made.
Note:
This holding statement ensures transparency and compliance while we work collaboratively to complete the project deliverables.
Benefits Realised
Data is currently being collected, analysed and reviewed and will be updated in due course.
Publications
No publications are planned outside of this holding statement.
GB-NON-12988 | May 2026
West Midlands Cancer Alliance (WMCA) SACT (Systemic Anti-Cancer Therapy) demand and capacity project.
Holding Statement – Collaborative Working Outcome Summary
Project Title
West Midlands Cancer Alliance (WMCA) SACT (Systemic Anti-Cancer Therapy) demand and capacity project.
Organisations involved
MSD UK
West Midlands Cancer Alliance (WMCA).
Trusts: University Hospitals Birmingham NHS Foundation Trust , University Hospitals Coventry and Warwickshire NHS Trust, South Warwickshire NHS Foundation Trust, George Eliot Hospital NHS Trust, Wye Valley NHS Trust, Worcestershire Acute Hospitals NHS Trust, The Shrewsbury and Telford Hospital NHS Trust, University Hospitals North Midlands NHS Trust, The Dudley Group NHS Foundation Trust, Sandwell and West Birmingham NHS Trust, Walsall Healthcare NHS Trust, The Royal Wolverhampton NHS Trust.
Summary
This 12-month collaborative working project was delivered in partnership with West Midlands Cancer Alliance (WMCA) and NHS Trusts across the region (listed above). The project focused on developing a robust, data‑led understanding of Systemic Anti‑Cancer Therapy (SACT) demand and capacity, to support service optimisation and sustainability across WMCA.
The objectives of this collaboration were as follows:
- Visibility of current and future capacity and demand (workforce and chair capacity) within SACT delivery sites across the cancer alliance.
- Optimised treatment and utilisation of services across at SACT delivery sites.
- Improved patient experience during SACT delivery across SACT delivery sites.
- Improve cancer waiting times to begin SACT in line with national guidance.
- Standardisation of practices in SACT delivery.
Using MSD’s Capacity Insights Tool (CIT), data was gathered from NHS SACT units and used to model current and future service needs, identifying gaps and opportunities to optimise delivery. Reports and insights were shared and reviewed at facilitated workshops and scenarios captured to improve patient access and efficiencies were explored.
This project supported NHS England’s (NHSE) 2024/2025 directive for biannual SACT capacity reviews and responds to rising demand, complex treatments, and workforce pressures. Outcomes include improved visibility of service capacity, optimised treatment scheduling, and reduced waiting times. Trusts considered action plans based on data-led insights, with progress followed up and potential impact captured.
Next Steps
- Partner organisations will align on the data and associated outcomes.
- Once agreed, the outcome summary will be finalised and submitted for certification.
- We will update stakeholders promptly as progress is made.
Note:
This holding statement ensures transparency and compliance while we work collaboratively to complete the project deliverables.
Benefits Realised
Data and outcomes are currently being reviewed and will be updated in due course.
Publications
No publications are planned outside of this holding statement.
GB-NON-12960 | May 2026
Wessex Cancer Alliance (WCA) and NHS Trusts Systemic Anti-Cancer Therapy (SACT) Day Unit Capacity Insights project
Project Title
Wessex Cancer Alliance (WCA) and NHS Trusts Systemic Anti-Cancer Therapy (SACT) Day Unit Capacity Insights project
Organisations involved
- Wessex Cancer Alliance
- MSD
- University Hospital Southampton NHS Foundation Trust
- University Hospitals Dorset NHS Foundation Trust
- Portsmouth Hospitals University NHS Trust
- Hampshire Hospitals NHS Foundation Trust
- Dorset County Hospital NHS Foundation Trust
- Isle of Wight NHS Trust
Summary
The aim of this project was to derive data-led insights on the demand and resource capacity of each Trust’s SACT Day Unit (SDU) to deliver existing and future treatment across Wessex Cancer Alliance using MSD’s capacity insights model (CIT). The visibility of the data, when added to narrative from day unit staff, could then support decision making that optimised SACT services to ensure timely access for patients to treatment with minimal waiting times.
The insights from the model highlighted gaps and opportunities, including quick wins, within the services’ ability to deliver current and future SACT.
Facilitated meetings, led by the Cancer Alliance SACT Project team and supported by MSD enabled a review of the insights report at each Trust and a discussion on potential modelled scenarios that, if implemented by SDUs, could address current/future service gaps. The data-led decisions were incorporated into a broader plan for Trust Cancer Services to influence and facilitate changes in line with both national, regional and local cancer strategy.
Project Objectives:
- Working together with SDU staff, scope the capacity and demands of each Trust’s SDUs by completing Capacity and Demand audit to show their current chair and workforce capacity to deliver vs their current demand, i.e., existing demand now, in 12 months and beyond.
- Communicating Trust specific modelled demand and resource requirement reports to 6 NHS Trusts within Wessex.
- An overarching regional report was disseminated to all key stakeholders including NHS Trusts, Treatment Board, Integrated Care Boards (ICB), SACT Clinical Advisory Group (CAG) and SACT Nurses Site Specific Group (SSG) to support decision making on SACT resource planning.
- Predict likely changes in demand and required service development to meet those changes to improve SACT capacity and, in so, reduce the 31-day cancer wait times for first and follow-up treatment.
- Reduce the frequency and impact of deferrals and delays to improve efficiency and patient experience.
- Understand and facilitate delivery of local action plans with each Trust based on the results of the project
- Develop an SDU performance dashboard that visualises the Trusts’/MSD report data, hosted by WCA, accessible to relevant Trust cancer services and the key clinical stakeholders.
Outputs/Take-homes from the project (May 2026)
- Clear regional evidence base across 6 acute Trusts (13 SACT units), establishing a validated baseline for chair capacity, SACT nurse workforce, activity mix, delays and deferrals.
- Current capacity is already under strain
- Local chair and workforce shortfalls persist despite apparent regional balance.
- 101 commissioned SACT chairs vs ~90 required for current demand, masking unit-level deficits where demand exceeds capacity.
- 6 of 13 SACT units are operating beyond sustainable capacity.
- Future SACT delivery is at risk: 5-year modelling shows that without intervention
- By 2030: regional shortfall of –31 SACT chairs and –24.5 WTE SACT nurses.
- Within 5 years, demand is projected to exceed current capacity in 10 of 13 SACT units.
- Workforce is the critical constraint
- SACT nurse shortages identified (71 WTE available vs 66 WTE required).
- Greatest pressures at UHS Hamwic House and Portsmouth HODU.
- Pathway inefficiencies identified (deferrals and delays)
- Delayed starts consume ~231 extra chair hours/week, reducing productive capacity and worsening experience.
- ~655 patients/week are delayed on the day of treatment.
- ~256 appointments/week are deferred across the region.
- 500 chair hours/week lost.
- Delayed starts consume ~231 extra chair hours/week, reducing productive capacity and worsening experience.
- Service reconfiguration could increase SACT capacity by relocating non‑SACT activity in some units
- Some SACT units deliver moderate volumes of non‑SACT care.
- System-level recommendations to support decision-making
- Trusts: modelled service redesign, workforce optimisation, scheduling, and non‑SACT reconfiguration.
- Increase chair and workforce capacity and improve efficiency.
- Expand subcutaneous SACT.
- Expand “SACT closer to home” delivery.
- Wessex Cancer Alliance: regional SACT leadership (CAG workplan including workforce innovation and digital dashboards).
- Optimise scheduling, reduce delays/deferrals, and redesign non‑SACT pathways.
- Support homecare for “SACT closer to home” models.
- Provides commissioners and system leaders with a robust case for investment, redesign and workforce planning across Wessex.
- Trusts: modelled service redesign, workforce optimisation, scheduling, and non‑SACT reconfiguration.
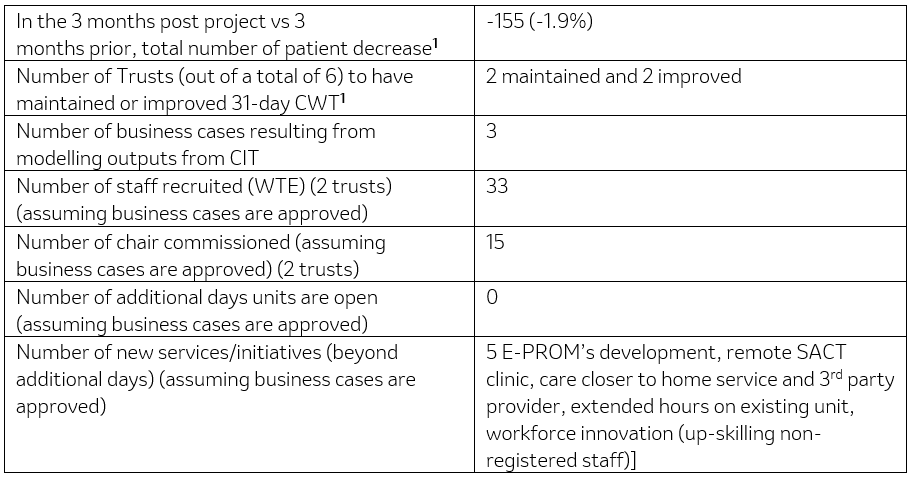
Proposed long term benefits
Patient Benefits
- Put patients first by improving patient experience and outcomes.
- Extra capacity in the service some of which will be delivered closer to home
- Shorter waiting times, increased access to treatment and improved outcomes
NHS Benefits
- WCA has greater insight into SACT services regionally, with a focus on gaps between capacity and demand. This helps direct resource planning.
- Align with WCA values
- Shared insight, learning and cross fertilisation of approaches in optimising SACT services
- Identification and improvement in the management of current inefficiencies across the SACT pathway
- Timely access for patients to SACT treatment in line with national targets.
MSD Benefits
- Enhanced reputation for MSD through partnership work.
- Better understanding of SACT treatment services.
- This collaboration may have enabled more patients to access innovative treatments in line with NICE guidance which may or may not have included MSD medicines
Lessons learnt
- Enable earlier action through shared baseline data (agree action-plan ownership, next steps and accountability with Trust leaders after each demand/capacity cycle).
- Don’t underestimate manual data burden Manual collection/cleansing alongside e-prescribing increases workload and variability; simplify requirements early and automate where possible.
- Align scope, definitions and success measures early
Shared definitions (capacity, delays, deferrals, lost/recoverable capacity) improve confidence, support consistent interpretation, and reduce rework across Trusts. - Clinical engagement builds credibility
1:1 engagement and iterative validation with Trust teams improved data quality, interpretation, and ownership. - Regional totals can mask unit-level risk
Moving from aggregate reporting to unit-level variation made findings more actionable. - Workforce is the binding constraint (not chairs)
Chair growth must be matched by investment in SACT nursing, pharmacy, aseptics and prescribers. - Activity mix matters as much as volume. Non‑SACT activity within SACT units can materially reduce available capacity.
- Future-demand modelling shifted the conversation. Five‑year modelling moved discussions from firefighting to strategic planning, strengthening the case for earlier commissioner involvement in workforce and service redesign.
- Tailor language and presentation for system audiences. Clearer explanations of data requirements and processes improved engagement, legitimacy and report usefulness.
Funding & Resources
This project is a shared commitment from Wessex Cancer Alliance (University Hospitals Southampton NHS Foundation Trust) and MSD
Publications
No intended publications.
References:
- NHS England Monthly CWT Statistics » 2025-26 Monthly Cancer Waiting Times Statistics (September-November 2025 vs September-November 2024) (Accessed: April 2026).
GB-NON-12957 | May 2026
The Christie Post Operative Clinic Pilot for Cancer Patients
Project Title
The Christie Post Operative Clinic Pilot for Cancer Patients
Organisations involved
MSD (UK) Ltd and The Christie NHS Foundation Trust
Summary
This was an eighteen‑month pilot at The Christie NHS Foundation Trust to test a dedicated, nurse‑led post‑operative clinic for renal and melanoma cancer patients. The pilot introduced a separate post‑operative pathway, supported by a Cancer Nurse Specialist (CNS) team and consultant oversight, to manage follow‑up care previously delivered through mixed clinics. Patient throughput and service activity were monitored throughout the pilot. The project end date was extended to February 2026 due to delays in patient recruitment
Benefits Realised
Benefits for Patients
Renal clinic
- Improved access to a dedicated post‑operative pathway: Renal post‑operative attendances increased from 113 in Q4 2024 (mixed clinic baseline) to 141 in Q4 2025 within the dedicated post‑operative clinic, indicating increased utilisation of the nurse‑led pathway. ²
- Proactive nurse‑led support between clinic visits: During the pilot, 51 nurse‑led telephone or email contacts were recorded for renal post‑operative patients, with 9 (17.6%) resulting in escalation for further medical review or admission. ²
- High early satisfaction with the renal post‑operative clinic: All renal post‑operative patient survey respondents reported high satisfaction with their clinic experience. 5
| Survey Aspect | Response |
| Explanation of scan results/treatment plan | 5/5 (very satisfied) |
| Ability to discuss concerns within appointment time | Yes |
| Knowing who to contact with issues | Yes, issues managed effectively |
| Likelihood of recommending the clinic | 5/5 (very likely) |
Melanoma clinic
- Post‑operative support for melanoma patients: In 2024, 128 melanoma patients were supported through the post‑operative clinic pathway. ¹
- Nurse‑led monitoring and escalation: Across this cohort, 98 nurse‑led telephone contacts were recorded, with 36 contacts resulting in escalation for further investigation or urgent clinical review. ¹
- Qualitative evidence of emotional and practical support: Patient feedback highlights the CNS as a trusted and accessible point of contact, providing regular check‑ins, support with side‑effect management, coordination of investigations, and reassurance during post‑operative treatment decision‑making. ⁴
Renal & Melanoma clinic – common patient benefits
- Named and trusted point of contact: Patients across both pathways have access to a dedicated post‑operative nurse between clinic visits. ⁴ ⁵
- Early identification and escalation of clinical issues
Nurse‑led contact in both clinics supports timely identification of concerns and appropriate escalation to medical teams or acute services. ¹ ²
Benefits for NHS (The Christie)
Renal clinic
- Shift of follow‑up activity from mixed clinics: Between Oct–Dec 2024 and Oct–Dec 2025, mixed‑clinic renal attendances reduced from 1,142 to 1,020, while post‑operative clinic attendances increased from 113 to 141, indicating a transfer of activity into the dedicated post‑operative service. ²
- Nurse‑led telephone triage supporting safe care delivery: Renal post‑operative telephone and email contacts (51 recorded) functioned as a structured triage pathway, with appropriate escalation for patients requiring further review. ²
Melanoma clinic
- Structured management of post‑operative melanoma follow‑up: In 2024, the post‑operative clinic managed follow‑up care for 128 melanoma patients through a defined nurse‑led pathway, including proactive monitoring and escalation where required. ¹
- Support for timely clinical decision‑making: Routine nurse‑led follow‑up and escalation of concerns enables earlier intervention and supports safer ongoing management of melanoma patients post‑surgery. ¹⁴
Renal & Melanoma clinic common NHS benefits
- Improved service planning and governance: Both services routinely capture data on clinic utilisation, nurse‑led contacts, escalation activity, and patient and staff experience, supporting service oversight, capacity planning, and future business case development. ² ³ ⁵
Following the successful pilot and business case approval, both nurse posts have been appointed on a full‑time basis, supporting continuation of the service.
Benefits for MSD
- Enhanced reputation through collaborative working: The project strengthened MSD’s reputation as a collaborative partner supporting service innovation and improved patient care within the NHS.
- As a pharmaceutical manufacturer of oncology medicine, an indirect result could be that MSD may see use of their NICE approved medicines.
Funding & Resources
This project was funded through a shared contribution between The Christie NHS Foundation Trust and MSD (UK) Ltd. The total project cost was £60,000.
Lessons learnt
- Separating post‑operative from metastatic pathways improves clinic flow and patient experience.
- Dedicated CNS or key‑worker support is critical for timely investigations, escalation, and continuity of care.
- Standardised discharge templates and coordinated CNS relationships reduce duplication and delays.
- Routine data capture enables responsive service optimisation and sustainability planning.
References
- Melanoma Patient counts (Data on File)
- KPI dashboard (Data on File)
- Staff survey responses (Data on File)
- Melanoma Feedback from patients (Data on File)
- Post-operative Renal Clinic Patient Survey (Data on File)
GB-NON-12870 | April 2026
Cheshire& Merseyside SACT Capacity, Demand & Utilisation Project
Project Title
Cheshire & Merseyside SACT Capacity, Demand & Utilisation Project
Organisations involved
- MSD UK
- Cheshire & Merseyside Cancer Alliance
Summary
The outcome summary for the Cheshire & Merseyside Systemic Anti-Cancer Therapy (SACT) Capacity, Demand & Utilisation Project is currently pending due to finalisation and alignment between parties. We remain committed to publishing the full results once the required information is received and validated.
For background, the aim of this project was to provide data-led insights into demand and capacity of SACT delivery in current and future services for Cheshire & Merseyside Cancer Alliance. This analysis was conducted using MSD’s capacity insights tool (CIT) and aims to support optimisation of cancer treatment services to ensure timely access to optimal treatment. The insights from the model highlighted gaps and opportunities within the services’ ability to deliver current and future SACT. Facilitated capacity workshops (led by MSD) enabled a review of the insights report and a discussion on potential modelled scenarios that, if implemented by SACT day units, could address current/future service gaps. The data-led decisions were incorporated into an action plan for Trust cancer services to influence and implement changes. The intent was for SACT delivery service capacity to improve (measured via a second report from CIT) and therefore allow for more timely access to optimal treatments for Cheshire & Merseyside Cancer Alliance.
This project is a 12-month collaboration between MSD UK and Cheshire and Merseyside Cancer Alliance.
Next Steps:
- Partner organisations will align on the data and associated outcomes.
- Once agreed, the outcome summary will be finalised and submitted for certification.
- We will update stakeholders promptly as progress is made.
Note:
This holding statement ensures transparency and compliance while we work collaboratively to complete the project deliverables.
Benefits Realised
Data is currently being collected, analysed and reviewed and will be updated in due course.
Publications
No publications are planned outside of this holding statement.
GB-NON-12843 | April 2026
East of England Cancer Alliance (EOECA) SACT (Systemic Anti-Cancer Therapy) demand and capacity project
Project Title
East of England Cancer Alliance (EOECA) SACT (Systemic Anti-Cancer Therapy) demand and capacity project.
Organisations involved
East of England Cancer Alliance (EOECA).
Trusts: Norwich and Norfolk University Hospitals NHS Foundation Trust, James Paget University Hospitals NHS Foundation Trust, Queen Elizabeth Hospital King’s Lynn NHS Foundation Trust, North West Anglia NHS Foundation Trust, Cambridge University Hospitals NHS Foundation Trust, West Suffolk NHS Foundation Trust, East Suffolk and North Essex NHS Foundation Trust, Milton Keynes University Hospital NHS Trust, Bedfordshire Hospitals NHS Foundation Trust, East and North Hertfordshire NHS Trust, Princess Alexandra Hospital NHS Trust, Mid and South Essex NHS Foundation Trust.
Summary
This 12-month collaborative working project was delivered in partnership with East of England Cancer Alliance (EOECA) and NHS Trusts across the region (listed above). The project focused on developing a robust, data‑led understanding of Systemic Anti‑Cancer Therapy (SACT) demand and capacity, to support service optimisation and sustainability across EOECA. Using MSD’s Capacity Insights Tool (CIT), data was gathered from NHS SACT units and used to model current and future service needs, identifying gaps and opportunities to optimise delivery. Reports and insights were shared and reviewed at facilitated workshops and scenarios captured to improve patient access and efficiencies were explored.
This project supported NHS England’s (NHSE) 2024/2025 directive for biannual SACT capacity reviews and responds to rising demand, complex treatments, and workforce pressures. Outcomes include improved visibility of service capacity, optimised treatment scheduling, and reduced waiting times. Trusts considered action plans based on data-led insights, with progress followed up and potential impact captured.
Outcomes
The process of modelling demand and capacity service data at a Trust level identified common themes across all NHS Trusts:
- Based on the current trajectories of expected growth in demand, SACT capacity in EOECA will not meet operational requirements within a range of 1 to 5 years, with Trusts already experiencing demand that is greater than available capacity, indicated by the modelling work undertaken.
- Current state findings indicate 57% of Trusts in EOECA are operating at or above chair capacity.
- Current state findings indicate 86% of Trusts in EOECA are operating at or above workforce capacity.
This project has laid the foundation for positive future intended outcomes. Examples of services/initiatives explored during feedback discussions:
- Homecare service expansion (e.g., developing protocols for future NICE-approved subcutaneous treatments, SACT @ Home models, community chemo bus/mobile unit service, expanding service delivery outside hospital units).
- Optimise oral SACT pathway / collection (e.g. Pharmacy led service to reduce burden on SACT unit staff, freeing up capacity to focus on in-chair treatments).
- Dedicated PICC line, supportive therapies, and rapid subcutaneous chairs (ringfencing activity to improve planning, capacity, and patient safety).
- Digital: Update & reconfigure relevant scheduling and/or prescribing software (optimises chair utilisation and scheduling, supporting capacity planning and reducing patient delays).
- Teleclinics for patient fit-to-attend confirmations (reducing delays and deferrals, giving pharmacy services more time to prepare treatments).
- Health & Wellbeing nurse roles (reducing patient anxiety, reducing delays & deferrals, acting as a safety net for under-resourced medical workforce).
- SACT unit co-ordinator roles (essential for improving operational efficiencies of SACT units – improves capacity management, reduces patient delays).
- Pre-chemo checks (nurse-led clinic), improves planning and reduces pharmacy delays.
- Centralised pre-assessments and scheduling improvements (building additional time into patient attendance for pharmacy preparation).
- Non-SACT activity/supportive therapies moved out of units (freeing up capacity for core chemotherapy services).
- Establish SACT best practice forums (internal and externally sharing best practice & learnings from local SACT units).
- Review SACT unit opening times and explore capacity sharing between Trust units/hospitals (flexible contracts), capacity sharing in scope to optimise capacity across the week/busier periods/emergencies.
Metrics
Table 1 below summarises metrics captured during the project, derived from CIT discussions and supporting data analysis, comparing February 2025 to a February 2024 baseline period.
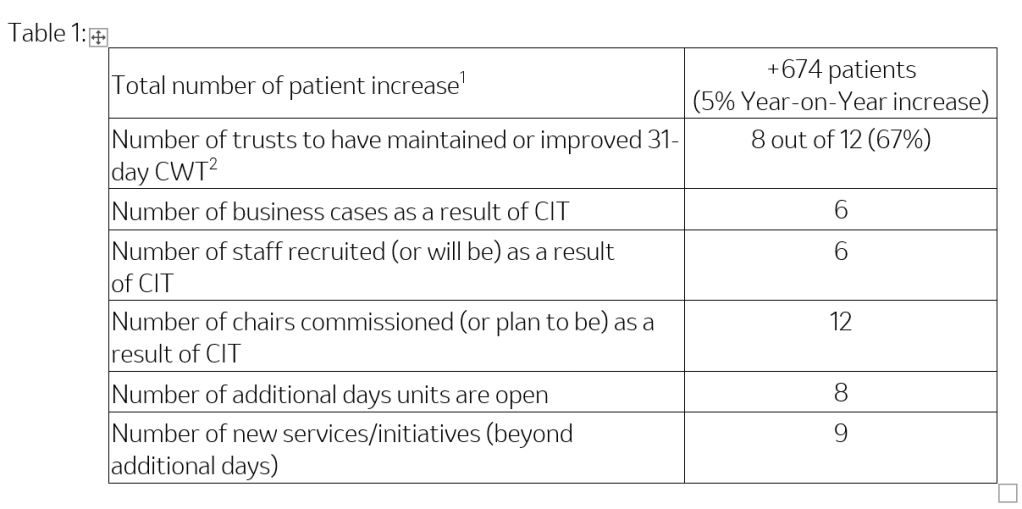
Benefits Realised
Patient Benefits:
- Through this collaboration, approximately 14,5021 patients have been impacted through improved SACT service delivery across East of England Cancer Alliance (EOECA).
- Patients experienced SACT services undergoing improvement initiatives to reduce treatment delays.
- Maintained or improved patient access and outcomes in line with 31-day Cancer Waiting Times (CWT) standard.
- Reducing anticipated delay to treatment as demand rises to 2030.
- Reducing avoidable ‘on the day’ deferrals i.e. not due to acute onset of illness arising from SACT.
NHS Benefits:
- 8 out of 12 Trusts maintained or reported an increase in its achievement of the 31-day CWT standard, with a corresponding decrease in the number of pathway breaches when comparing July-September 2025 to a baseline period of July-September 2024.
- East of England Cancer Alliance (EOECA) and associated Trusts in the region, now have an overview of their SACT services demand and capacity status to help deliver a more regional approach; supporting planning and optimising resource use, highlighting where additional resource is required.
- Improving treatment capacity to fulfil current and future demand.
- Data led evidence generated to support service improvement, initiatives, and business case activity for increased staffing and additional SACT chairs commissioning.
- Opportunity to further explore treatment in the community models, aligning to the new national cancer plan and potentially releasing VAT savings for the NHS organisation.
MSD Benefits:
- Enhanced reputation for MSD through partnership work.
- Better understanding of treatment services and improvement opportunities.
- As a pharmaceutical manufacturer of oncology medicines, an indirect result of improved treatment capacity may be that MSD see more appropriate usage of their NICE approved medicines.
Funding & Resources
This was a project with shared time commitment from East of England Cancer Alliance & MSD.
Lessons learned
- Labour intensive data collection (manual, paper-based) was time consuming and potentially prone to error; adopting electronic solutions and focusing data collection on essential areas would reduce workload and improve accuracy.
- NHS clinician/admin time and resource to conduct detailed analysis such as time and motion studies is extremely limited which can act as a headwind to a project’s completion. However, when this activity is conducted it can yield valuable insights when analysed through an appropriate modelling capability such as the Capacity Insights Tool (CIT).
- Full stakeholder engagement and formal governance from the start of the project enabled successful planning and implementation.
- Consistent use of the CIT data template and CIT tool across all units facilitated direct comparability.
Publications
No intended publications.
References
1 National Disease Registration Service (NDRS) NHS England (NHSE) SACT Activity Dashboard.
https://digital.nhs.uk/ndrs/data/data-outputs/cancer-data-hub/sact-activity
(Accessed: March 2026)
2 NHS England Cancer Waiting Times (CWT). Statistics » Cancer Waiting Times
(Accessed: March 2026)
GB-NON-12650 | March 2026
East Midlands Cancer Alliance (EMCA) SACT (Systemic Anti-Cancer Therapy) demand and capacity project.
Project Title
East Midlands Cancer Alliance (EMCA) SACT (Systemic Anti-Cancer Therapy) demand and capacity project
Organisations involved
East Midlands Cancer Alliance (EMCA).
Trusts: Nottingham University Hospitals NHS Trust, Sherwood Forest Hospitals NHS Trust, United Lincolnshire Hospitals NHS Trust, Northampton General Hospital NHS Trust, Kettering General Hospital NHS Trust, University Hospitals of Derby & Burton NHS Trust, University Hospitals of Leicester NHS Trust.
Summary
This 12-month collaborative working project was delivered in partnership with East Midlands Cancer Alliance (EMCA) and NHS Trusts across the region (listed above). The project focused on developing a robust, data‑led understanding of Systemic Anti‑Cancer Therapy (SACT) demand and capacity, to support service optimisation and sustainability across EMCA. Using MSD’s Capacity Insights Tool (CIT), data was gathered from NHS SACT units and used to model current and future service needs, identifying gaps and opportunities to optimise delivery. Reports and insights were shared and reviewed at facilitated workshops and scenarios captured to improve patient access and efficiencies were explored.
This project supported NHS England’s (NHSE) 2024/2025 directive for biannual SACT capacity reviews and responds to rising demand, complex treatments, and workforce pressures. Outcomes include improved visibility of service capacity, optimised treatment scheduling, and reduced waiting times. Trusts considered action plans based on data-led insights, with progress followed up and potential impact captured.
Outcomes
The process of modelling demand and capacity service data at a Trust level identified common themes across all NHS Trusts:
- Based on the current trajectories of expected growth in demand, SACT capacity in EMCA will not meet operational requirements within a range of 1 to 5 years, with Trusts already experiencing demand that is greater than available capacity, indicated by the modelling work undertaken.
- Current state findings indicate 43% of Trusts in EMCA are operating at or above chair capacity.
- Current state findings indicate 100% of Trusts in EMCA are operating at or above workforce capacity.
This project has laid the foundation for positive future intended outcomes. Examples of services/initiatives explored during feedback discussions:
- Homecare service expansion (e.g., developing protocols for future NICE-approved subcutaneous treatments, SACT @ Home models, community chemo bus/mobile unit service, expanding service delivery outside hospital units).
- Optimise oral SACT pathway / collection (e.g. Pharmacy led service to reduce burden on SACT unit staff, freeing up capacity to focus on in-chair treatments).
- Dedicated PICC line, supportive therapies, and rapid subcutaneous chairs (ringfencing activity to improve planning, capacity, and patient safety).
- Digital: Update & reconfigure relevant scheduling and/or prescribing software (optimises chair utilisation and scheduling, supporting capacity planning and reducing patient delays).
- Teleclinics for patient fit-to-attend confirmations (reducing delays and deferrals, giving pharmacy services more time to prepare treatments).
- Health & Wellbeing nurse roles (reducing patient anxiety, reducing delays & deferrals, acting as a safety net for under-resourced medical workforce).
- SACT unit co-ordinator roles (essential for improving operational efficiencies of SACT units – improves capacity management, reduces patient delays).
- Pre-chemo checks (nurse-led clinic), improves planning and reduces pharmacy delays.
- Centralised pre-assessments and scheduling improvements (building additional time into patient attendance for pharmacy preparation).
- Non-SACT activity/supportive therapies moved out of units (freeing up capacity for core chemotherapy services).
- Establish SACT best practice forums (internal and externally sharing best practice & learnings from local SACT units).
- Review SACT unit opening times and explore capacity sharing between Trust units/hospitals (flexible contracts), capacity sharing in scope to optimise capacity across the week/busier periods/emergencies.
Metrics
Table 1 below summarises metrics captured during the project, derived from CIT discussions and supporting data analysis, comparing February 2025 to a February 2024 baseline period.
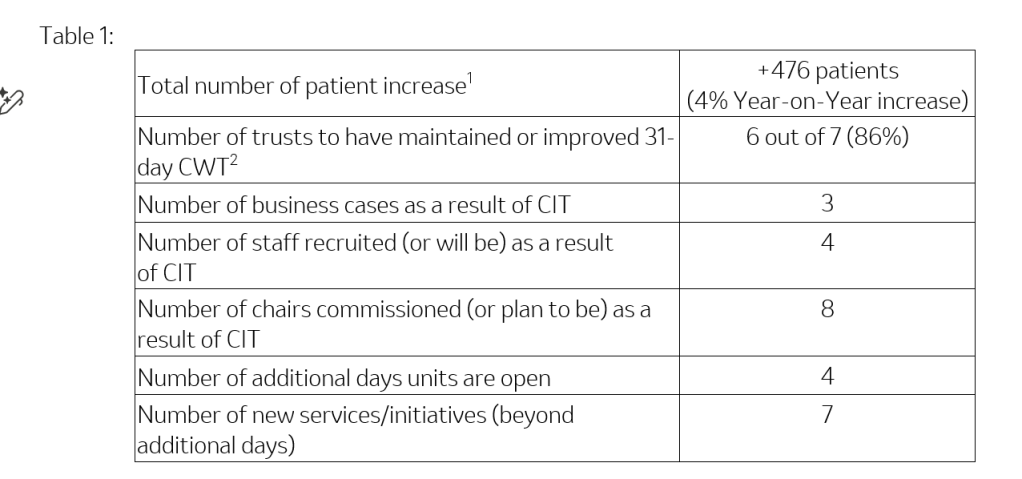
Benefits Realised
Patient Benefits:
- Through this collaboration, approximately 11,0711 patients have been impacted through improved SACT service delivery across East Midlands Cancer Alliance (EMCA).
- Patients experienced SACT services undergoing improvement initiatives to reduce treatment delays.
- Maintained or improved patient access and outcomes in line with 31-day Cancer Waiting Times (CWT) standard.
- Reducing anticipated delay to treatment as demand rises to 2030.
- Reducing avoidable ‘on the day’ deferrals i.e. not due to acute onset of illness arising from SACT.
NHS Benefits:
- 6 out of 7 Trusts maintained or reported an increase in its achievement of the 31-day CWT standard, with a corresponding decrease in the number of pathway breaches when comparing July-September 2025 to a baseline period of July-September 2024.
- East Midlands Cancer Alliance (EMCA) and associated Trusts in the region, now have an overview of their SACT services demand and capacity status to help deliver a more regional approach; supporting planning and optimising resource use, highlighting where additional resource is required.
- Improving treatment capacity to fulfil current and future demand.
- Data led evidence generated to support service improvement, initiatives, and business case activity for increased staffing and additional SACT chairs commissioning.
- Opportunity to further explore treatment in the community models, aligning to the new national cancer plan and potentially releasing VAT savings for the NHS organisation.
MSD Benefits:
- Enhanced reputation for MSD through partnership work.
- Better understanding of treatment services and improvement opportunities.
- As a pharmaceutical manufacturer of oncology medicines, an indirect result of improved treatment capacity may be that MSD see more appropriate usage of their NICE approved medicines.
Funding & Resources
This was a project with shared time commitment from East Midlands Cancer Alliance & MSD.
Lessons learned
- Labour intensive data collection (manual, paper-based) was time consuming and potentially prone to error; adopting electronic solutions and focusing data collection on essential areas would reduce workload and improve accuracy.
- NHS clinician/admin time and resource to conduct detailed analysis such as time and motion studies is extremely limited which can act as a headwind to a project’s completion. However, when this activity is conducted it can yield valuable insights when analysed through an appropriate modelling capability such as the Capacity Insights Tool (CIT).
- Full stakeholder engagement and formal governance from the start of the project enabled successful planning and implementation.
- Consistent use of the CIT data template and CIT tool across all units facilitated direct comparability.
Publications
No intended publications.
References
1 National Disease Registration Service (NDRS) NHS England (NHSE) SACT Activity Dashboard.
https://digital.nhs.uk/ndrs/data/data-outputs/cancer-data-hub/sact-activity
(Accessed: March 2026)
2 NHS England Cancer Waiting Times (CWT). Statistics » Cancer Waiting Times
(Accessed: March 2026)
GB-NON-12649 | March 2026
SACT Demand and Capacity planning across hospitals within Peninsula Cancer Alliance
Project Title
SACT Demand and Capacity planning across hospitals within Peninsula Cancer Alliance
Organisations involved
Peninsula Cancer Alliance and MSD
Summary
This 12-month project, in partnership with Peninsula Cancer Alliance, assessed the demand and capacity of systemic anti- cancer treatment (SACT) services across a total of 8 day units within 4 NHS Trusts using MSD’s Capacity Insights Tool (CIT). The tool was used to model current and future service needs, identifying gaps and opportunities to optimise delivery. Insights were shared and reviewed at facilitated workshops and scenarios to improve patient access and efficiency were explored. The project supported NHS England’s 24/25 directive for biannual SACT capacity reviews and responds to rising demand, complex treatments, and workforce pressures. Outcomes include improved visibility of service capacity, optimised treatment scheduling, and reduced waiting times. Trusts implemented action plans based on data-led insights, with progress followed up and impact captured.
Benefits Realised
Patient Benefits
- Approximately 10,125 patient lives 1 have been positively impacted through improved SACT service delivery across the Peninsula Cancer Alliance
- Patients experienced SACT services undergoing improvement initiatives to shorten delays
- Patients’ outcomes may potentially improve as a result
NHS Benefits
75% of Provider Trusts managed increased patient volumes without compromising on achievement of the 31-day CWT standard when comparing Aug-Oct 2025 to a baseline period of Aug-Oct 24 2:
- 75% of Trusts maintained their 100% achievement of the 31-day cancer waiting time standard from decision to treat to 1st definitive treatment
- 3 out of 4 Trusts managed an increase in the cancer patient volume within their SACT treatment services without a corresponding rise in the number of pathway breaches
- 2 of these Trusts managed an increase in patient volume while maintaining their 31-day CWT performance
3 Trusts have been able to develop business cases based on the insights from this work. 2 of which seek to gain funding for dedicated ultrasound resources for their day units to enable better venous access techniques. 1 of which seeks to gain funding for 3.5 WTE SACT Nurses to improve workforce capacity.
The process of modelling demand and capacity service data at a Trust and Unit level identified common themes across all Trusts:
- Based on the current trajectories of expected growth in demand, SACT capacity within the Peninsula will not meet operational requirements within a range of 1 to 5 years, with 2 Units already seeing chair demand that is greater than available chair capacity, and 4 day units seeing workforce demand that is greater than workforce availability according to the modelling work undertaken
- Variable issues identified for continuous improvement: venous access and cannulation support using dedicated ultrasound transducer within SACT services; opening of ‘inactive’ chairs and diverting demand to satellite locations; need to recruit additional workforce leading to business case creation from modelling insights generated
MSD Benefits
- Enhanced reputation for MSD through partnership work
- Better understanding of treatment services and improvement opportunities
- As a pharmaceutical manufacturer of oncology medicines, an indirect result of improved treatment capacity may be that MSD see more appropriate usage of their NICE/SMC approved medicines
Funding & Resources
This was a project with shared time commitment from Peninsula Cancer Alliance & MSD
Lessons learnt
NHS clinician time and resource to conduct detailed analysis such as time and motion studies are extremely limited which can act as a headwind to a project’s completion. However, when this activity is conducted it can yield valuable insights when analysed through an appropriate modelling capability such as the capacity insights tool
Publications
No intended publications
1 NHSE Cancer Waiting Times Data for total patients treated across all 4 provider Trusts from January 2025 to December 2025 filtered for 31 Day Treatment Standard and SACT Treatment Modality
2 NHSE Cancer Waiting Times Data – provider Trust achievement of the 31-day CWT standard when comparing Aug-Oct 2025 to a baseline period of Aug-Oct 2024
GB-NON-12726 | March 2026
Surrey & Sussex Cancer Alliance Urgent Suspected Lung, Breast & Gynae Cancer Pathology Pathway Development Programme (PDP)
Project Title
Surrey & Sussex Cancer Alliance Urgent Suspected Lung, Breast & Gynae Cancer Pathology Pathway Development Programme (PDP)
Organisations involved
SURREY & SUSSEX CANCER ALLIANCE and MERCK SHARP & DOHME (UK) LIMITED
Summary
The Purpose of this Project was to optimise the Urgent Suspected Cancer (USC) Pathology Pathways in Lung, Breast & Gynaecology from biopsy taken in theatre to final report being written up and made available by the trusts that are served by the Berkshire & Surrey Pathology Service (BSPS) within Surrey & Sussex Cancer Alliance (SSCA). This project aimed to improve pathway service quality, efficiency, and productivity. The desired outcome was to see an improvement and less variation in turnaround times for histopathology in Lung, Breast and Gynaecology across the 4 trusts which in turn could positively impact the 28 days Faster Diagnostic Standard and 31-day treatment target Cancer Waiting Time targets.
This was planned to be delivered collaboratively by both partners via the co-development and engagement in pathway mapping workshops and gap analysis workshops to identify barriers and blocks. Based on the findings from the gap analysis, both partners would subsequently work towards implementing plans of the identified areas of improvement and commit to measuring the outcomes of the project and disseminating these outcomes within 6 months of the project completion. The project intended to run for approximately 12 months.
Benefits Realised
Due to sickness within Surrey and Sussex Cancer Alliance and MSD, the project did not complete in the pre-defined timescales, and therefore no benefits were realised as a result.
Funding & Resources
This was a project with shared time commitment from Surrey and Sussex Cancer Alliance & MSD.
Lessons learnt
Creation of a formal contingency plan should any of the key stakeholders and MSD project manager have time capacity issues or their organisation suffers significant disruption which affects stakeholder capacity during the implementation of the project.
SSCA and MSD will work towards commencing a new project during 2026.
Future engagement will include pre-booked regular touchpoint meetings between MSD and key stakeholders. It is anticipated that in the event of the frequent postponement of these meetings, MSD and the stakeholder will meet separately to discuss any required action to ensure the project timeline is not disrupted.
Publications
There are no intended publication plans outside of this Outcomes Summary.
GB-NON-12591 | March 2026
Lancashire & South Cumbria SACT Capacity, Demand & Utilisation Project
Project Title
Lancashire & South Cumbria SACT Capacity, Demand & Utilisation Project
Organisations involved
Lancashire & South Cumbria Cancer Alliance & MSD Trusts: Lancashire Teaching Hospitals NHS Foundation Trust, Blackpool Teaching Hospital NHS Foundation Trust, East Lancashire Hospitals NHS Trust, The University Hospitals Morecambe Bay NHS Foundation Trust.
Summary
The aim of this project was to provide data-led insights into demand and capacity of systemic anti-cancer treatment (SACT) delivery in current and future services for Lancashire & South Cumbria Cancer Alliance (LSCCA). This analysis was conducted using MSD’s capacity insights tool (CIT) which aims to support optimisation of cancer treatment services to ensure timely access to optimal treatment.
The insights from the model highlighted gaps and opportunities within the services’ ability to deliver current and future SACT. We organised capacity workshops, led by MSD together with the LSCCA project manager, and carried out a two-week time and motion study across all eight units. Through observing the units and gathering data, we were able to review the insights report and discuss possible modelled scenarios. If these scenarios are adopted by SACT day units, they could help address current and future gaps in service provision.
The data led decisions have since been incorporated into an action plan for LSCCA working in partnership with the Lancashire & South Cumbria Integrated Care Board (ICB) and Trusts to influence and implement changes. The intent is for SACT delivery service capacity to improve and therefore allow for more timely access to optimal treatments whilst demand for SACT treatments continues to grow. The project ran over 12 months and has led to an implementation plan 2026-2030 led by LSCCA.
Benefits Realised – Projected Improvements1
Patient Benefits
- Reducing anticipated delay to treatment as demand rises to 2030
- Reducing delays in treatment on SACT units
- Reduction in avoidable ‘on the day’ deferrals e. not due to acute onset of illness arising from SACT
- Improved patient experience including incorporating Electronic Patient Reported Outcome Measures (ePROMS)
NHS Benefits
- Increased treatment capacity to fulfil current and future demand
- Lancashire & South Cumbria Cancer Alliance has an overview of their SACT services demand and capacity status to help deliver a more regional approach recommending a plan optimising resource use and highlighting where additional resource is required
- Opportunity to release VAT savings in treatment in the community models; otherwise unexplored in LSC
MSD Benefits
- Enhanced reputation for MSD through partnership work
- Better understanding of treatment services and improvement opportunities
- As a pharmaceutical manufacturer of oncology medicines, an indirect result of improved treatment capacity may be that MSD see more usage of their NICE approved medicines
Findings and Observations:
Current state1,2
- LSC SACT units are ~20.2 Whole Time Equivalent (WTE) understaffed to meet current demand
- Performance for the 31 days cancer wait time (CWT) is above national standard of 96%, regularly >98% showing that SACT units are working under significant pressure to maintain performance; this was already acknowledged as a fragile service
- The utilisation of chairs within LSC SACT units has been calculated at 72%.
Recommendations2:
Next Steps:
- LSCCA working in partnership with key stakeholders have planned to deliver the following to keep up with the anticipated demand for SACT services
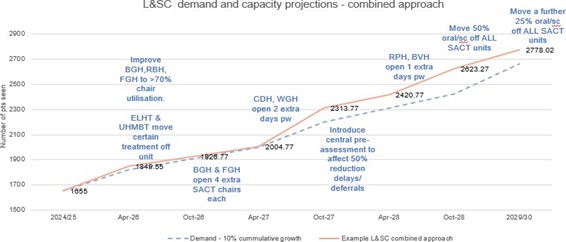
Burnley (BGH) Blackburn (RBH) Blackpool (BVH) Chorley (CDH) East Lancs Hospital Trust (ELHT) Preston (RPH) Westmorland (WGH) Furness (FGH) Lancaster (RLI) University Hospital of Morecombe Bay Trust (UHMBT)
Next steps:
- Implementing local improvements in 3 of the 8 units with <70% chair utilisation including scheduling processes, patient communication, staffing and drug reconstitution on unit
- Plans in each unit to grow number of chairs/days open as estate allows, working with Trusts to deliver this at Trust level
- Centralising pre-first treatment SACT assessment (this is currently being managed in-chair and contributing to the delay to treatment stats)
- Centralising pre-visit triage SACT assessment (this is currently being managed in-chair and contributing to the delay to treatment stats)
- Incorporating ePROMS into centralised triage to prioritise patients in need of support and expedite when necessary for acute intervention. Improving patient experience and two-way communication
- Piloting a treatment in the community hub and spoke model, to increase capacity for high volume, high cost drugs funded by the VAT saving this generates
- Aseptic improvements in capacity and procurement to allow improved capacity to keep up with the demand increase on SACT units
- Any next steps will not be addressed as part of this collaborative working project
Funding & Resources
This was a project with shared time commitment from Lancashire & South Cumbria Cancer Alliance & MSD
Lessons learnt (LSCCA statements)3
Findings and Recommendations
- Extensive manual, paper-based data collection was burdensome and prone to error; adopting electronic solutions and focusing data collection on essential areas would reduce staff workload and improve accuracy.
- Full stakeholder engagement and formal governance from the start enabled successful programme launch and collaborative planning, though limited staffing reduced on-site presence and affected stakeholder perceptions.
- Consistent tool use across all units facilitated direct comparability, but reports were often overly detailed and required significant reformatting to be accessible for stakeholders.
References
- Lancashire & South Cumbria CIT dashboard report
- SACT Transformation steering group presentation
- LSC CIT Finding, recommendation quote and email consent
GB-NON-12242 | November 2025
SACT Demand and Capacity planning across hospitals within SWAG Cancer Alliance
Project Title
SACT Demand and Capacity planning across hospitals within SWAG Cancer Alliance
Organisations involved
SWAG Cancer Alliance and MSD
Summary
This 12-month project, in partnership with SWAG Cancer Alliance, assessed the demand and capacity of systemic anti- cancer treatment (SACT) services across six NHS Trusts using MSD’s Capacity Insights Tool (CIT). The tool was used to model current and future service needs, identifying gaps and opportunities to optimise delivery. Insights were shared and reviewed at facilitated workshops and scenarios to improve patient access and efficiency were explored. The project supported NHS England’s 24/25 directive for biannual SACT capacity reviews and responds to rising demand, complex treatments, and workforce pressures. Outcomes include improved visibility of service capacity, optimised treatment scheduling, and reduced waiting times. Trusts implemented action plans based on data-led insights, with progress followed up and impact captured.
Benefits Realised
Patient Benefits
- Approximately 13,114 patient lives1 have been positively impacted through improved SACT service delivery across the SWAG Cancer Alliance
- Patients experienced SACT services undergoing improvement initiatives to shorten delays
- Patients’ outcomes may potentially improve as a result
NHS Benefits
Provider Trusts managed increased patient volumes without compromising on achievement of the 31-day CWT standard when comparing May-July 2025 to a baseline period of March-April 24 2:
- All Trusts continued to exceed the 31-day cancer waiting time standard of 96% from decision to treat to 1st definitive treatment
- 4 out of 6 Trusts managed an increase in the cancer patient volume within their SACT treatment services
- 1 Trust saw an increase in its achievement of the 31-day standard, with a corresponding decrease in the number of pathway breaches
- A further 2 two trusts managed an increase in patient volume while maintaining their 31-day CWT performance
The process of modelling demand and capacity service data at a Trust level identified common themes across all Trusts:
- Based on the current trajectories of expected growth in demand, SACT capacity in SWAG will not meet operational requirements within a range of 1 to 5 years, with 3 Trusts already seeing demand that is greater than available capacity according to the modelling work undertaken
- Variable issues identified for continuous improvement: venous access, variation in scheduled vs actual appointment duration; a need to streamline appointment cancellation processes; provision of patient information and education to avoid late arrival;, cancellations and delays to treatment.
This project has laid the foundation for future intended outcomes:
- Gloucester Hospitals Foundation Trust have indicated they will use the project data and insights as a foundation for a future workforce business case looking at the expansion of the SACT Nursing workforce and also channelling more appropriate activity through the mobile cancer unit to alleviate capacity pressures in the day unit.
- Somerset Foundation Trust will use the project data and insights to confirm scheduling accuracy and look to areas to improve efficiency of delays/bookings
- Salisbury Foundation Trust will use the project data and insights to inform an improvement project around SACT day unit operational efficiency
- University Hospitals Bristol and Weston will use project data and insights to provide valuable evidence for proposals to expand the satellite units and provide additional chair base and capacity, as well as transitioning to subcutaneous formulations for further operational efficiencies and improve their process for the management of deferrals
MSD Benefits
- Enhanced reputation for MSD through partnership work
- Better understanding of treatment services and improvement opportunities
- As a pharmaceutical manufacturer of oncology medicines, an indirect result of improved treatment capacity may be that MSD see more appropriate usage of their NICE/SMC approved medicines
Funding & Resources
This was a project with shared time commitment from SWAG Cancer Alliance & MSD
Lessons learnt
NHS clinician time and resource to conduct detailed analysis such as time and motion studies is extremely limited which can act as a headwind to a project’s completion. However, when this activity is conducted it can yield valuable insights when analysed through an appropriate modelling capability such as the capacity insights tool
Publications
No intended publications
1 NHSE Cancer Waiting Times Data for total patients treated across all 6 provider Trusts from August 2024 to July 2025 filtered for 31 Day Treatment Standard
2 NHSE Cancer Waiting Times Data – provider Trust achievement of the 31-day CWT standard when comparing May-July 2025 to a baseline period of March-April 24
GB-NON-12089 | October 2025
Earlier Diagnosis of Ovarian and Kidney Cancer in Brighton and Hove
Project Title
Earlier Diagnosis of Ovarian and Kidney Cancer in Brighton and Hove
Organisations Involved
Sussex Integrated Care Board and MSD UK
Summary
This project aimed to address the apparent inequalities in Brighton and Hove around mortality rates for ovarian and renal cancers identified within the 2023 Cancer Joint Strategic Needs Assessment (JSNA) report and the Inequalities in mortality involving common physical health conditions England: Office for National Statistics, Aug 2023. The collaboration aimed to increase early detection and diagnosis of people with ovarian and renal cancers.
MSD worked in partnership with Sussex Integrated Care Board (ICB) to provide project management support to the implementation and evaluation of work to address this.
The project ran for 12 months with the final evaluation 6 months post-implementation of all project related activities.
Project Objectives
- Reduce the current inequalities by increasing early detection and diagnosis rate of patients with ovarian and renal cancers.
- Support the provision of enhanced education and training support to increase knowledge regarding symptom awareness in undiagnosed ovarian and kidney cancer, including a clear definition of the issues that currently contribute to later stage diagnosis in ovarian and renal cancers to all GPs, Nurses and Pharmacists, and other Allied Healthcare Professionals (AHPs) who make clinical assessments of patients in primary care.
- Drive increased uptake in the use of the already integrated Ardens Pro and Ardens-EMIS ‘Urgent Suspected Cancer (USC) Referral Form templates for gynaecological and urology cancers.
- Produce a report / case study capturing the learnings that make a case for further prioritisation of ovarian and renal cancer symptom awareness and education on the best practice diagnostic steps to be adopted at scale across the Surrey and Sussex Cancer Alliance (SSCA).
Benefits Realised
Benefits Impact to Patient
- Increased awareness of the risk factors and early symptoms of ovarian cancer via Patient Group radio programme and television interviews on BBC Sussex.
- Increased awareness of the risk factors and early symptoms of ovarian and kidney cancers through Healthy Living Pharmacies via video case studies, posters and patient information leaflets developed and distributed to patients in Brighton and Hove during Campaign Awareness months (September 2024 and March 2025).
- During the duration of the project, there was an increase in the number of patients diagnosed with ovarian and kidney cancer at an earlier stage versus the pre-project baseline 6-month period.
- For ovarian cancer, of the cancers that were staged, the percentage diagnosed at stage 1 and 2 increased by 3.8% to 43.8% (7 cancers – University Hospital Sussex Somerset data).
- For kidney cancer, of the cancers that were staged, the percentage diagnosed at stage 1 increased by 12.6% to 57.8% (26 cancers – University Hospital Sussex Somerset data).
Benefits to the NHS Partners/Integrated Care System (ICS)
- Development of a truly multi-disciplinary project team creating stronger relationships across primary and secondary care, public health, university, cancer alliance and health innovation network to advance the above work.
- Training for health care professionals in the ‘earlier recognition and diagnosis of ovarian and kidney cancer’ via:
- Improved support materials, networked across partner media to advance earlier symptom recognition at presentation by GP’s through e.g.
- Ovarian Cancer Updated Advice and Guidance statements on ‘Suspected Gynaecological referral forms’ – May 2024 and Target Ovarian ‘Cancer Facts and Stats’ (Surrey and Sussex Cancer Alliance website).
- Healthy Living Pharmacy Training materials: new booklets and videos to increase staff awareness of the signs and symptoms of ovarian and kidney cancer, requested and shared with NHSE.
- Pharmacy Staff Training – All 15 Brighton Healthy Living Pharmacies received training on early symptom awareness at presentation for:
- Ovarian cancer – September 24
- Kidney cancer – November 24
- GP Training webinars: – recorded and shared across the ICS as an ongoing learning resource, and made available through the ICB /Sussex Training Hub:
- Diagnosing Ovarian Cancer Earlier in Primary Care Sussex webinar 18-Oct-24: 23 on the day recipients in total.
- Diagnosing Kidney Cancer Earlier in Primary Care Sussex webinar 24-Jan-25: 9 on the day recipients in total.
- Ovarian cancer diagnosed during the project:
- A decrease in the number of patients diagnosed in the 6 months to April 2025 (29) versus the pre-project baseline 6-month period to March 2024 (40).
- Of the cancers that were staged, the percentage diagnosed at stage 1 and 2 increased from 40.0% at baseline (19 cancers) to 43.8% (7 cancers), an increase of 3.8% (University Hospital Sussex Somerset data).
- Kidney cancers diagnosed during the project:
- An increase in the number of patients diagnosed in the 6 months to April 2025 (67) versus the pre-project baseline 6-month period to March 2024 (40).
- Of the cancers that were staged, the percentage diagnosed at stage 1 increased from 45.2% at baseline (14 cancers) to 57.8% (26 cancers), an increase of 12.6% (University Hospital Sussex Somerset data).
- Improved support materials, networked across partner media to advance earlier symptom recognition at presentation by GP’s through e.g.
Benefits to MSD
- Enhanced reputation and legitimacy from working collaboratively in partnership with Sussex Integrated Care System (ICS).
- A better understanding of customer and patient needs in ovarian and kidney cancer.
- Increased understanding of the earlier detection and diagnosis of both ovarian and kidney cancer in the primary care setting as well as the challenges to current inter-service diagnostic provision within secondary care.
- Identification of possible solutions to enable early symptom identification by GPs and Pharmacists that may help reduce the historic later stage diagnosis in Brighton and Hove.
- This collaboration may have enabled more patients to access innovative treatments in line with NICE guidance which may or may not have included MSD medicines.
Funding and Resources
This was a project with shared time commitment from Sussex ICS and MSD.
Project Stakeholder Quote
“The joint working of this project produced some extremely valuable outputs for both cancers that can be utilised for several years and in several settings. The work also demonstrated how we can act upon JSNA findings as a catalyst for action, rather than using them for reference and information. This should align well with the future direction of ICBs and ‘strategic commissioning’, together with the ability to influence integrated action at a neighbourhood level.
It was a pleasure to have direct project management support from MSD”.
Mark Hannigan – Project Lead
Lessons Learnt:
- Access to diagnostic and staging data held in secondary care continues to be a challenge. This is exacerbated by the ovarian specific patient data being bundled within the gynaecological cancers’ ‘dataset’ and kidney specific data being bundled within the urological cancers’ ‘dataset’. Boundaries to access still exist with partners, e.g. limitations of access by Sussex University to the Sussex Integrated Dataset.
- The Integrated Care Board (ICB) were unable to engage Brighton and Hove Primary Care Networks (PCNs) to participate in the project work around earlier diagnosis (education) and diagnosis (e.g. enabling Ardens-Pro) to increase the awareness and skills necessary to use the new systems for earlier detection including, diagnostics, referral pathways, safety netting, advice, and guidance. Primary Care engagement in proactive additional activities remains challenging both contractually and in terms of time commitment.
- Against current work pressures and priorities, engaging primary care healthcare professionals to complete a survey on symptom awareness and diagnostic practices was a challenge, including the use of Ardens-Pro decision support tool to aid diagnosis and increase current detection rates for the two tumour sites. This meant that we did not collect sufficient data to demonstrate whether the training interventions had an impact on earlier identification of both cancers
- The ICB communications team/platform has a relatively weak/limited reach amongst the many communications directed at primary care to engage health care professionals to attend the two earlier diagnosis webinars within their protected learning time.
- Co-production remains a very effective tool in developing materials and resources. Where co-production includes wider health and care partnerships, charities and patients.
- The engagement of academic colleagues and support significantly enhances the findings where statistical outlier activity is highlighted as the focus of a project.
Publications
No publications are planned outside of this Outcomes Summary.
GB-NON-11457 | June 2025
MDT Reform Project – Cheshire and Merseyside Cancer Alliance
Project Title
MDT Reform Project – Cheshire and Merseyside Cancer Alliance
Organisations Involved
Cheshire and Merseyside Cancer Alliance c/o The Clatterbridge Centre NHS Foundation Trust.
Summary
The project aimed to provide project management support for two days per month for 8 months to Cheshire and Merseyside Cancer Alliance in supporting the implementation of multi-disciplinary team (MDT) reform. The project focussed on implementing MDT triage at The Countess of Chester Hospitals (COCH), and initially focussed on breast, skin, urology and colorectal pathways. The pre-MDT triage standards would be developed by the Trusts in conjunction with the Cancer Alliance, and MSD would support in implementing the standards across the four MDTs. MSD were only involved in the implementation and not the development of the standards.
The project aimed to drive forward national recommendations and guidance for MDTs to deliver an improved MDT process. This would be done through improving the effectiveness of cancer MDTs; ensuring streamlined processes/ standards of care pathways are developed and implemented to make the best use of clinical time and resources.
The project aimed to benefit patients from an accelerated review through the pathway, either in MDT or outside an MDT, using pre-agreed protocols for those less urgent or severe cases. As a result, treatment decisions are likely to be made earlier than in current practice, thus facilitating accelerated treatment timelines, less anxiety for patients waiting for this decision, and potentially patients deemed as fit for additional treatment options.
Benefits Realised
Due to changes in key stakeholders at MSD, the project encountered delays. Furthermore, competing priorities at the Countess of Cheshire hospital arose due to these delays, which meant that the project did not complete in the pre-defined timescales, and therefore no benefits were realised as a result.
Funding & Resources
This was a project with shared time commitment from Countess of Chester Hospital, Cheshire and Merseyside Cancer Alliance & MSD.
Lessons Learnt
Creation of a formal project contingency plan to ensure project continuity considering any key stakeholder movement and/or sickness.
Publications
There are no intended publication plans outside of this Outcomes Summary.
GB-NON-11392 | May 2025
Virtual SACT pre-assessment using the MySunrise App across Devon and Cornwall Hospitals
Project Title
Virtual SACT pre-assessment using the MySunrise App across Devon and Cornwall Hospitals
Organisations Involved
Peninsula Cancer Alliance, Technical Health Limited and MSD Ltd
Summary
MSD worked in partnership with Peninsula Cancer Alliance (PCA) and Technical Health Limited to provide project management support to implement and evaluate a virtual pre-assessment service for cancer patients waiting to undergo systemic anti-cancer therapy (SACT). The new service involved a digital health intervention through the already established MySunrise App.
Our aims were to:
- Provide a virtual option for patients to undergo SACT pre-assessment prior to coming into hospital to receive treatment.
- Release clinical capacity and healthcare professional time.
- Provide a better patient experience from not having to travel to their hospital for a clinic appointment that can be conducted virtually.
- Create an evidence base and list of benefits realised to enable wider adoption of virtual pre-assessment.
Patient Benefits
- We believe that through implementing this project we have positively impacted 6135 patients over a 12-month period in 2024 across Cornwall and Torbay Hospitals. This is assuming a 47% uptake of virtual preassessment versus the in-person option. This has been calculated using NHSE cancer waiting times data1.
- Improved patient experience resulting from a total of:
- 2760 miles of travelling distance saved (20 miles per patient average)
- 92 hours of travelling time saved (40 mins per patient average)
- 602Kg of CO2 reduced in total (4.36Kg CO2 saved per patient)
- £8.35 saved per patient on travel costs (estimated)
- Improved patient experience as virtual pre-assessment option allows families to join appointment.
- Staying at home reduces infection risk .
NHS Benefits
- Virtual SACT pre-assessment has been established as preferred patient engagement route in Cornwall and Torbay Hospitals
- The following results were combined from operational running of SACT virtual pre-assessment service across November 2023 – May 2024 in Torbay and across January 2024 – April 24 in Cornwall:
- 196 patients had SACT virtual pre-assessment for treatment within project duration across the Peninsula region, comprised from:
- 58 patients in Cornwall
- 138 in Torbay
- Capacity improved as 162 SACT Nurse hours saved from implementing virtual pre-assessment across the Peninsula region (£3,268 cost equivalent).
- Clinical room capacity released as 138 hours of estate time saved.
- 196 patients had SACT virtual pre-assessment for treatment within project duration across the Peninsula region, comprised from:
- SACT treatment rates increased by 28% in Torbay from February (883 patients) to June 2024 (1131 patients).
MSD Benefits
- An opportunity to contribute towards the development of innovative SACT pre-assessment services across all cancer pathways, gaining experience of service development initiatives.
- Collaborating on a project that creates a use case to positively impact patients through implementation of virtual SACT pre-assessment across other geographies and speaks to the government’s 10-year health plan for the NHS to prioritise shifting from analogue to digital2.
- This collaboration may ultimately have enabled more patients to access innovative treatments in line with NICE guidance which may or may not have included MSD medicines.
Funding & Resources
This project was a shared funding commitment from Peninsula Cancer Alliance, Technical Health Limited & MSD. The total project cost was £10,000.
Lessons learnt
- Engage stakeholder organisations around the evaluation metrics at the start of the project and agree on data collection plan.
- Ensure regular touch points across all stakeholder organisations to ensure clarity of expectation, review of implementation and outcomes, and best practice sharing.
Publications
SACT virtual pre-assessment poster exhibited at UKONS 2024
- NHSE Cancer Waiting Time Provider data extract 2024
- King’s Fund – The 10 Year Health Plan
GB-NON-11361 | May 2025
Lancashire Teaching Hospital Trust Skin and Urology Diagnostic Histopathology Pathway Development Programme
Project Title
Lancashire Teaching Hospital Trust Skin and Urology Diagnostic Histopathology Pathway Development Programme
Organisations involved
MSD-UK
Lancashire Teaching Hospital Trust
Summary
The collaboration aimed to optimise the Skin and Urology diagnostic histopathology pathway at Lancashire Teaching Hospitals Trust to improve the service quality, efficiency, and productivity. This aimed to support achieving the Royal College of Pathologists Key Performance Indicator (RCPath KPI) of 90% of samples reported within 10 calendar days, impacting NHS Cancer Waiting Time targets including the 28-day Faster Diagnosis Standard (FDS) within six months. The project sought to enhance patient care by improving turnaround times for histopathology in Skin and Urology diagnostics through pathway mapping, gap analysis, optimisation workshops and pathway improvement implementation.
Benefits Realised1
Through implementation of MSD’s Pathway Development Programme with Lancashire Teaching Hospital NHS Foundation Trust Histopathology team, MSD provided project management support to assess the turnaround times of their histopathology pathway from receipt of sample to diagnostic result reporting. A gap analysis was carried out and the areas below were identified as areas of improvement to reduce the turnaround times (TAT) reduction in line with the RCPath KPI (90% Sample received reported within 10 calendar days)2:
- Workforce – pathologist and biomedical scientist recruitment.
- Specimen reception area expanded
- Improve Histopathology requesting and labelling process.
- Training / education on request status and histopathology handbook
- New workflow and processes for specimen prioritisation, allocation, triage and reporting
Impact on turnaround time2

Fig 1
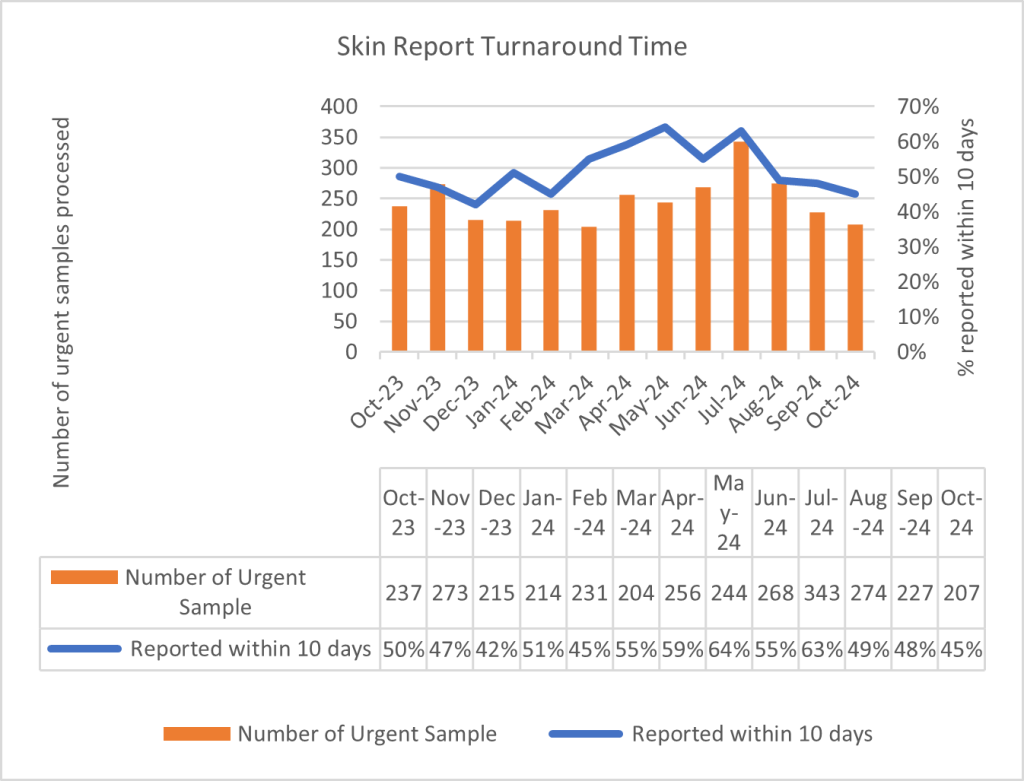
Fig 2
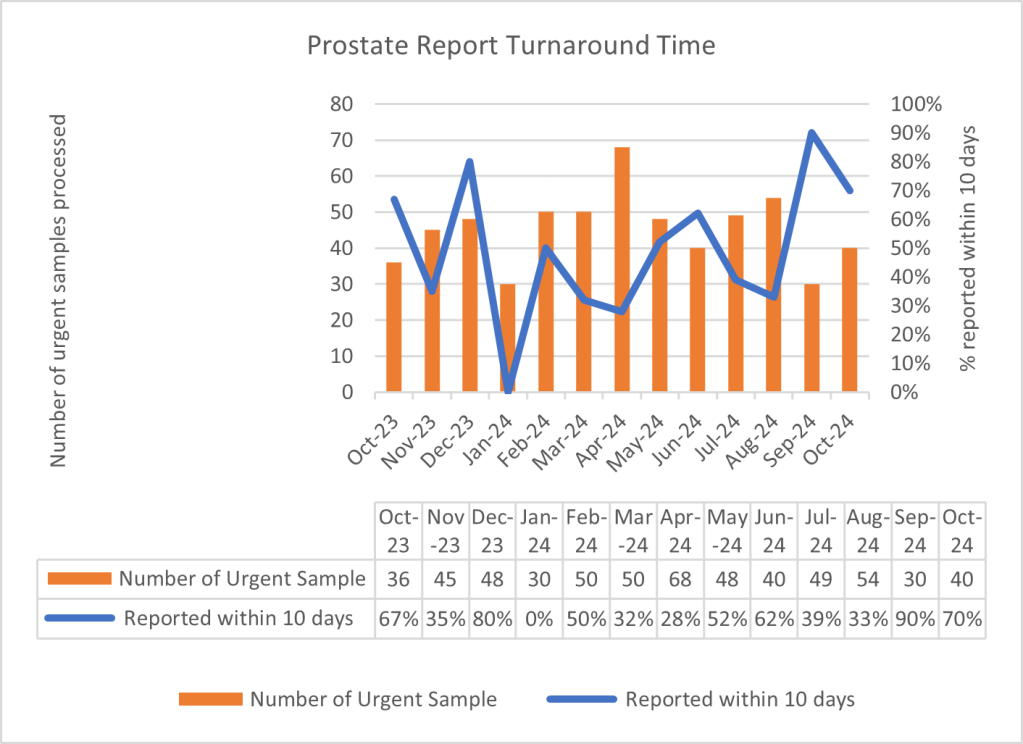
Fig 3
Patient benefits realised
- Faster diagnosis and shorter treatment pathway3: The reduction in turnaround times has enabled faster reporting of diagnosis of urology tumours from March 2024, when only 44% were being reported within 10 days, to 88% in October 2024 (Fig 1) which is crucial for timely and effective treatment. Unfortunately, a similar result was not displayed in the case of the skin pathway, where reporting of samples within 10 days decreased from 55% in March 2024 to 45% in October 20242.
NHS benefits realised
- The optimisation of the histopathology pathway has led to a more streamlined and efficient process. This was identified through the optimised pathway.
- Improved turnaround times: The project has demonstrated significant progress towards achieving the RCPath KPI of 90% reporting within 10 days for Urology, with July and August hitting the 90% target (Fig 1).
- Resource allocation: Efforts to recruit additional consultant pathologists have been made to ensure adequate resources are available to meet the increased demand.
- Optimised Service Delivery3: The optimised pathway has facilitated earlier histopathology requests and more appropriate requesting for Skin and Urology tumour patients. Dedicated, internal communications were shared as part of the new workflow process being implemented. Qualitative feedback has suggested the service have seen an improvement in request appropriateness.
- Pathway improvements: The implementation of new workflows and the recruitment of additional staff have contributed to the optimisation of service delivery.
MSD benefits realised
- Better understanding of patient needs: Gathering and analysing data provide MSD with better understand the impact of histopathology on time to treatment patients with Skin / Urology cancer.
- Improved reputation: Strengthening partnerships and showcasing successful collaborations have improved MSD’s reputation, this has led to more discussions with the Cancer Alliance about future projects.
- As a pharmaceutical manufacturer of oncology medicines, an indirect result of an improved pathway may be that MSD see more appropriate usage of their NICE/SMC approved medicines.
Funding & Resources
This project is a shared contribution of time between Lancashire Teaching Hospital Trust, Lancashire & South Cumbria Cancer Alliance and MSD.
NHS Stakeholder quotes4
‘’The redesign and expansion of our cellular pathology specimen reception has allowed the operational flow to be improved dramatically. Segregation of specimen work streams has allowed efficient triage of the suspected cancer cases, reduced risk to patients and allowed timely interventions when requisition issues arise’’.
Recommendations:
- Need for Continuous Improvement: The project highlighted the necessity of ongoing improvement and adaptation to meet changing needs and circumstances.
- Effective Communication: The continuation of clear and effective communication will prove crucial for the smooth implementation of new workflows and processes in future.
- Resource Allocation Challenges: The challenges in recruiting appropriate candidates underscored the need for strategic resource planning and management.
- Workflow Optimisation: The use of colour-coordinated bags is the only outstanding action recommended to implemented. This will continue to demonstrate the importance of optimising processes to enhance efficiency and communication.
- Improved turnaround time: A review of the process for Skin is needed to understand why similar improvements were not achieved and to identify lessons that can be applied from the Urology success.
- Workflow Mapping: following the successful implementation of a new workflow design in conjunction with new workspace; the service requires to process map the new workflow and capture all points in a tissue sample journey.
Publications
There are no intended publication plans outside of this Outcomes Summary.
References
- LTHTR HistoPath – Highlight report
- Impact on turnaround time data
- Optimised pathway map
- Stakeholder Email – Quote
GB-NON-11246 | April 2025
Immuno-oncology nurse-led service
Project Title
Immuno-oncology nurse-led service
Organisations involved
University College London Hospitals (UCLH) NHS Foundation Trust
MSD
Objectives
- Establishment of a dedicated immuno-oncology (IO) nurse-led service to run for 12 months within the scope of the project, followed by a 3-month evaluation and publication to share the learns with peers and the wider NHS.
- Recruitment of an immunotherapy clinical nurse specialist (CNS) who will set up and lead IO service delivery.
- To secure funding through UCLH to continue the service after the project.
The service aimed to achieve the following:
- All new patients will have access to the educational element of the service including those that are changing therapy to immune checkpoint inhibitors (ICIs).
- Patients requiring additional management of ICIs will be flagged to the service and advice given will be tailored support to meet the patient’s needs and circumstances. Advice can be given either to the patient directly or via the managing clinician. The aim is that all eligible ICI patients believe they have been adequately educated and given the correct information to manage their treatment.
- All staff working with patients being treated with ICIs feel adequately upskilled to support patients through their journey, measured via questionnaire across nurses, pharmacists and oncologists.
Responsibilities of the Immunotherapy CNS:
- Management of the immuno-oncology service, including participation in clinics, meetings and follow up.
- Supporting the patient’s journey when on immuno-oncology treatment; specifically ICIs.
- Educating patients on their treatment, developing and providing patient information for a diverse population.
- Offering treatment support including advising treating clinicians on an individual case basis.
- Education of specific patient needs for staff including nurses, pharmacists and oncology clinicians and the wider HCPs involved in cancer patient care.
- Co-ordination of care, supporting patients, managing investigations and appointments as appropriate for patients on ICIs.
- Support innovation and improving services by adopting best practices.
- Ensuring adherence to best practice and local guidelines.
Summary
This collaboration supported the set up and development of an “immuno-oncology nurse-led service” for patients receiving ICIs. This involved UCLH recruiting an immunotherapy CNS to deliver and evaluate the nurse-led service for the duration of 12 months (an extension of 6 months was made to the project duration due to difficulties in recruiting for the immunotherapy CNS position; a variation agreement was used to document and formalise this extension). This service was supported by a specialist team at UCLH with project management from MSD and financial input from both parties.
The project demonstrated the value of an IO nurse-led service for patients who are treated with ICIs, reducing the variation of education and treatment support across different cancer types ensuring consistency throughout the trust.
Long term funding for the IO nurse service and immunotherapy nurse was achieved and a successful service was created that received 101 referrals from November 2023 to October 2024.
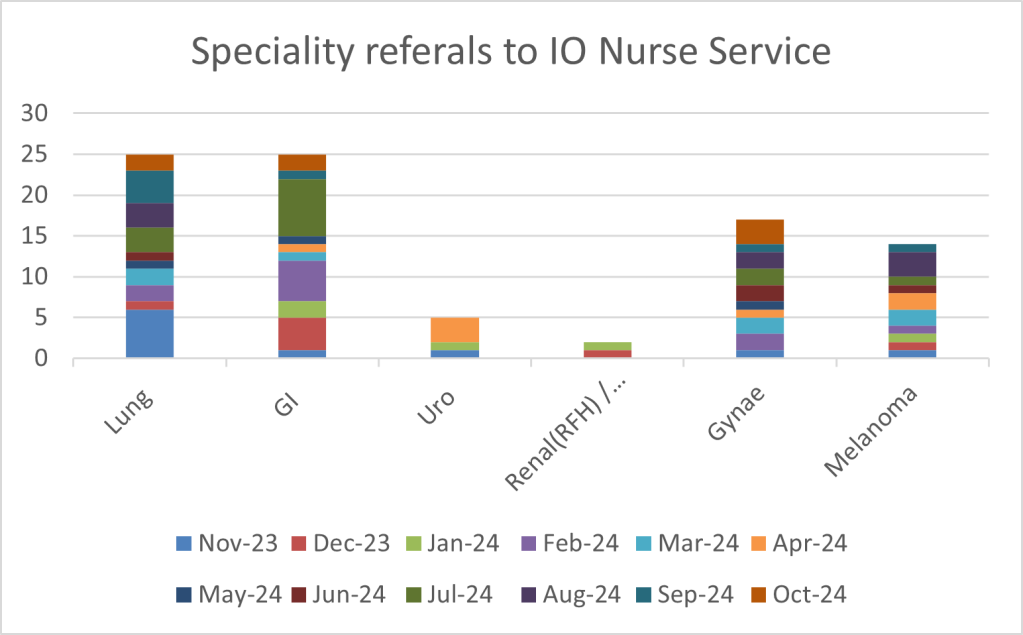
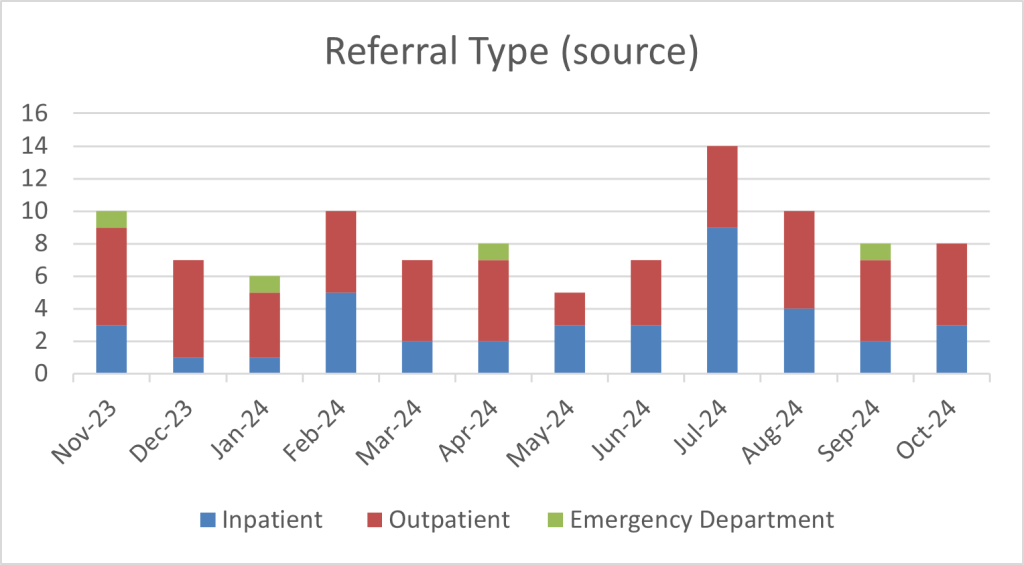
Graphs created from UCLH IO Nurse Referral Data, November 2023 – October 2024.
Patient survey – To assess the impact of the project on patients’ education and understanding of their ICI treatment, pre- and post- project surveys were conducted. Participation rose from 32 to 53 patients, reflecting increased engagement. Additionally, the percentage of patients willing to contact CNSs for significant bowel movements increased from 71% to 90.5%. There was also a notable increase in patients expressing their intention to seek help for worsening fatigue, rising from 44% to 66%. These findings underscore the project’s effectiveness in enhancing patient education and promoting proactive health-seeking behaviours.
The survey responses represented roughly a quarter of the 215 patients undergoing active ICI treatment. There has been a notable improvement in patients’ ability to manage their treatment, and this knowledge is anticipated to grow further with the upcoming CNS training and the dissemination of patient information resources about ICI treatment.
Staff survey – The primary objective of this survey is to evaluate the knowledge and confidence levels of staff regarding their understanding of ICI management. The response rate for this survey was lower compared to the previous one; however, all respondents reported having encountered ICIs in their clinical practice.
Overall, approximately 67% of participants reported being either very confident or somewhat confident in ICI management, up from 58% in the previous year.
Notably, the use of trust guidelines has risen significantly, with 100% of respondents now utilising them, compared to just 80% previously. Additionally, the number of reported opportunities for formal training has increased dramatically, from 7% last year to 82% this year, highlighting the positive impact of the role played by the immunotherapy CNS.
It is important to note that the survey did not receive any responses from junior doctors, pharmacists, or research nursing staff, and there were very few responses from ward or chemotherapy day care staff, despite them having participated in most training sessions led by the immunotherapy CNS. This underscores the necessity for greater involvement of tumour CNSs in training initiatives and the continuation of group training sessions to further enhance knowledge and confidence among the staff.
Benefits Realised
Benefits impact to patients
- Patients received a dedicated service and indirect point of contact to manage their treatment.
- Patient survey demonstrated marked improvements in patient’s basic knowledge of ICIs
- Further perceived improvement with publication of patient information sheets on ICIs.
- Better understanding of the difference between IO and chemotherapy.
Benefits to the NHS partners
- Continued funding of the IO service and dedicated immunotherapy CNS nurse role
- Improved confidence of staff managing patients on ICIs
- Consistent approach and education in ICI treatment management
- Internal study event – first ever Immunotherapy only study event, delivered by specialists, 92% of the attendees rated this as extremely useful.
- Tailored teaching sessions- feedback from 47 participants ranges from staff nurses, research teams and junior doctors showed improving confidence in managing ICIs after sessions
- Joint working with NCL partners, study events and conferences
Benefits to MSD
- Reputational benefit of working collaboratively with UCLH
- Improved management of ICI treatment due to increased education of staff and patients
- This collaboration may have enabled more patients to access innovative treatments in line with NICE guidance which may or may not have included MSD medicines
Funding & Resources
This project was a shared funding commitment from UCLH & MSD. The total project cost was £70,000.
Project Stakeholder Quote
”The MSD project has been very vital to develop and deliver a service which focuses on patient experience, patient safety, continuity of care and improved efficiency mainly through patient education and staff training. I am very optimistic that this service will improve patient safety and outcomes, increase awareness amongst staff, to prepare to equip them with the impact of the introduction of new immune checkpoint inhibitors. Thank you so much’’. – Ganga Sasidharan Pillai, Immunotherapy CNS at the University College London Hospitals NHS Foundation Trust
Lessons learnt
- Recruitment of the IO nurse position took significantly longer than anticipated which led to the project requiring a time extension.
- Having a network of these types of roles enables best practice sharing and continued improvement of these types of service throughout the NHS.
- The importance of understanding the success criteria and expertise required to navigate the process to secure continued funding of the IO service and IO CNS.
Publications
No intended publication plans outside of this Outcomes Summary.
GB-NON-11173 | April 2025
Immunotherapy Service Mapping in Swansea Bay and Hywel Dda University Health Boards
Project Title
Immunotherapy Service Mapping in Swansea Bay and Hywel Dda University Health Boards
Organisations involved
MSD, Swansea Bay University Health Board and Hywel Dda University Health Boards
Summary
Swansea Bay University Health Board (SBUHB) and Hywel Dda University Health Board (HDUHB) together as an immunotherapy service task group worked in collaboration with MSD UK to map out and baseline the current service for dealing with oncology patients that receive immunotherapy (IO).
SBUHB and HDUHB together form South West Wales cancer services with SBUHB being the hub for IO support. The two Health Boards wished to map out their current service with the aim of then enhancing and upskilling services across the hub and spoke service. It is anticipated that the project will be completed within 4 months. The project started in November 2023 and finished in March 2024.
Benefits Realised
- Through implementation of this project with the SBUHB and HDUHB teams, MSD provided project management support to assess the current state of the service for patients receiving IOs. The mapped pathway was used to provide a baseline which informed a subsequent business plan for providing a dedicated IO service across the two Health Boards. As a result of the project, the following benefits were realised:
Patient Benefits
- The aim of providing a dedicated IO service is for patients to receive better and more efficient care whilst receiving immunotherapy. This is the aim within the service redesign if the business case is successful. At this point the business case is in the final stages of development before submission.
- Patient experience was not measured as part of this project.
NHS Benefits
- This project provided the baseline for the development of a business case for a dedicated IO service. This business case will be submitted in the near future and if successful will hopefully provide:
- Potentially scalable service changes that could be rolled out across the two Health Boards
- The expansion of existing skill base within SBUHB and HDUHB.
- The ability to release capacity for existing expertise in SBUHB and HDUHB through the expansion of skills and services.
- Additionally, the project provided a detailed baseline and comparison of how IO patients receive care across the two Health Boards. This highlighted the need for a better understanding of the data and services across the two organisations due to acute oncology services having so many differences between the two Health Boards.
MSD Benefits
- Enhanced reputation of MSD through partnership work. Five NHS stakeholders completed a survey following the project and the results are shown below:
- Average score of 8.6/10 and 7.8/10 was scored for being satisfied with MSD’s collaborative working project and believed MSD contributed to their organisation’s cancer service pathway respectively
- Average score of 8.8/10 in the likelihood of working with MSD and the pharmaceutical industry in the future (up from 7/10 prior to the PDP)
- Quotes taken from the survey when questioned, “what has improved in your service/pathway following this collaborative working project” from NHS Stakeholders:
- “More clear in depth understanding the pathway and the way the service now and the points of improvement”
- When questioned on feedback of your experience of working in collaboration with MSD:
- “Great staff – helpful and also respectful of our clinical time. I would collaborate again.”
- I was unaware of MSD prior to this project, they have been really helpful and flexible finding the time to work around my shifts when I have been busy on the unit. They made discussions easy and straight forward and helped develop the pathways.”
- “Personable, clear and professional interactions with MSD
- Additionally, the project helped MSD to have a better understanding of IO patient needs
This collaboration may have enabled more patients to access innovative treatments in line with NICE guidance which may or may not have included MSD medicines.
Funding & Resources
This was a project with shared time commitment from Swansea Bay and Hywel Dda University Health Boards & MSD-UK.
Lessons learnt
- Communication is key between all parties which is especially important between departments to optimise decision making and establish the appropriate intervention for the patient as early as possible
- Regular touch points to ensure clarity of expectation and outcomes
- Clear plan of action and review within stakeholder meetings
- The care of patients receiving immunotherapy remain challenging. There are significant workforce challenges across Swansea Bay and Hywel Dda University Health Boards.
Publications
There are currently no plans to publish the outcomes of this project by the Health Boards as per the date of this summary.
GB-NON-10692 | December 2024
South Yorkshire & Bassetlaw Cancer Alliance Pan Tumour Pathology Pathway Development Project (PDP)
Project Title
South Yorkshire & Bassetlaw Cancer Alliance Pan Tumour Pathology Pathway Development Project (PDP)
Organisations involved
Sheffield Teaching Hospital NHS Foundation Trust, Barnsley Hospital Foundation Trust, The Rotherham NHS Foundation Trust, Doncaster and Bassetlaw Teaching Hospital Foundation Trust and Chesterfield Royal Hospital Foundation Trust
Summary
There is an opportunity in the South Yorkshire & Bassetlaw and Chesterfield area to optimise the pan-tumour pathology testing pathway to improve the service quality, efficiency, productivity, as well as patient experience. The desired outcome of this project was an improved pan-tumour histopathology molecular testing pathway within the regional trusts and demonstrate improvements of the relevant targets associated with head and neck/skin/breast/lung/gynaecological cancers, 28 day Faster Diagnostic Standard and the 31- and 62-day Cancer Waiting Time Targets. The project began on 1st October 2022 and finished on 30th September 2023.
Benefits Realised
Patient
- Improved timelines for patients to receive a cancer diagnosis.
- Improved timelines for patient access to personalised medicines.
NHS
- Greater understanding of regional workload for cancer histopathology services.
- Created engagement & collaboration across previously siloed histopathology laboratories.
- Consolidation of regional specialised cancer histopathology services.
- Sheffield Teaching hospitals approved to become a Regional Cellular Pathology Genomic Hub.
- Inhouse pilot of regional Lung NGS testing service resulted in a pathway reduction of 12 days (March 2024 data of NGS request to authorised result Inhouse Tat = 8 days v Referred TaT = 20 days)
- % of patients achieving 31-day CWT improved from 92% to 94% in the lung pathway, and from 78% to 86% in the head and neck pathway from July 2022 to December 2023.
- % of patients achieving 62-day CWT improved from 63% to 67% in the lung pathway, and from 74% to 78% in the breast pathway from July 2022 to December 2023.
- HER2 FISH testing planned to be brought inhouse in 2024 which has the potential to reduce pathways with associated HER2 FISH testing by 14 days
MSD
- Greater understanding of the complexity of molecular testing pathways which allow patients access to personalised systemic anti-cancer treatments.
- Enhanced reputation of MSD through partnership work.
- This collaboration may have enabled more patients to access innovative treatments in line with NICE guidance which may or may not have included MSD medicines.
Funding & Resources
This was a project with shared time commitment from South Yorkshire and Bassetlaw cancer Alliance & MSD.
Lessons learnt
- In depth stakeholder engagement plan is critical to ensure timely decisions can be made to enable service improvements are implemented as soon as possible.
- Ensure that NHS stakeholders build protected time into their working schedule so that the project milestones can be completed on time, and therefore eliminated the need for project overrun.
Publications
There are currently no plans to publish the outcomes of this project by the hospital trust as per the date of this summary.
GB-NON-10208 | September 2024
East Lancashire Hospitals NHS Trust Pan Tumour (Breast, Lung, Skin, Upper GI) Diagnostic Pathology Pathway Development Project (PDP)
Project Title
East Lancashire Hospitals NHS Trust Pan Tumour (Breast, Lung, Skin, Upper GI) Diagnostic Pathology Pathway Development Project (PDP)
Organisations involved
MSD-UK
East Lancashire Hospitals NHS Trust
Summary
A national optimal Pan Tumour (Breast, Lung, Skin, Upper GI) Diagnostic Pathology pathway has been created with the intention of improving patient experience through promoting quality cancer care and ensuring all (Pan Tumour (Breast, Lung, Skin, Upper GI) patients receive optimal cancer care. There is an opportunity in East Lancashire to optimise the Pan Tumour (Breast, Lung, Skin, Upper GI) Pathology pathway in line with the national optimal Pan Tumour (Breast, Lung, Skin, Upper GI) Diagnostic Pathology pathway to improve the service quality, service efficiency, productivity and patient experience. The desired outcome of this project is an improved Pan Tumour (Breast, Lung, Skin, Upper GI) Diagnostic Pathology pathway aligned with the national optimal pathway and achievement of the Pan Tumour (Breast, Lung, Skin, Upper GI) Diagnostic Pathology pathway turnaround times reduction, 28 day Faster Diagnostic Standard and the 31 and 62 day Cancer Waiting Time Targets. The project intends to run for approximately 6 months.
Benefits Realised
Due to significant and unanticipated capacity issues within East Lancashire Hospitals NHS Trust which needed to take priority, the outcome summary for the East Lancashire Hospitals NHS Trust Pan Tumour PDP has not been initiated and has been delayed.
Funding & Resources
This was a project with shared time commitment from University Hospitals of Birmingham (UHB) & MSD
Lessons learnt
Creation of a formal contingency plan should any of the key stakeholders and MSD project manager have time capacity issues, leave their post or their organisation during the implementation of the project.
Publications
No publications are planned.
GB-NON-09802 | August 2024
Clatterbridge Cancer Centre (CCC) Cancers of Unknown Primary (CUP) Pan Tumour Diagnostic Pathway Development Project (PDP)
Project Title
Clatterbridge Cancer Centre (CCC) Cancers of Unknown Primary (CUP) Pan Tumour Diagnostic Pathway Development Project (PDP)
Organisations involved
MSD-UK
Clatterbridge Cancer Centre
Summary
A national optimal CUP Pan Tumour Diagnostic pathway has been created with the intention of improving patient experience through promoting quality cancer care and ensuring all CUP patients receive optimal cancer care. There was an opportunity in Merseyside to optimise the CUP Pan Tumour diagnostic pathway in line with the national optimal CUP Pan Tumour pathway to improve the service quality, service efficiency, productivity, and patient experience. The desired outcome of this project was an improved CUP Pan Tumour Diagnostic pathway aligned with the national optimal pathway. This pathway does not fall under the 28-day Cancer Waiting Time targets so had to access on its own entity. The project intended to run for approximately 6 months.
Actions taken which resulted in the benefits realised
Patient benefits
- Optimised the CUP genomic pathway across systems by facilitating Whole Genomic Sequence (WGS) pathway and reducing turnaround time.
- Highlighted system-wide challenges in the CUP genomic pathway, such as delays in CT guided biopsy and lab processes.
- Established a Genomic Education group to support access to genomic resources and tools for patients and staff.
- Trialling innovation tag tracking Radio Frequency Identification Device (RFID) system for tissue specimens to avoid duplication of having to acquire the sample again and the stress to the patient due repeat the procedure.
- Created a tracker to capture turnaround time (TAT) and patient outcomes for the CUP pathway.
- Developed a patient story video to share with relevant leads and a wider audience to raise awareness of pathway changes.
- Developed a genetic form and communication plan to support easy access to genomic test results within the Clatterbridge Cancer Centre Meditech system to improve treatment rate and patients’ outcomes.
NHS Benefits1
During the six months of the optimised pathway project, 14 patients with CUP were eligible for WGS via the NHS test directory. 57% (n8) had an additional biopsy for WGS, 42 % (n6) did not have the WGS biopsy as they needed to start chemotherapy urgently, and the additional time to arrange biopsy would delay their treatment. At the time of this report (July 2024), 36% (n5) patients are still awaiting the results, it is difficult to present accurate data to demonstrate the turnaround time improvements in the pathway.
Following the improvements from this project multiple benefits can be actualised:
- The communication process with the lab teams has improved, there have been no further missing samples.
- There has been wider communication of the pathway and process, staff are now more aware of the differences in the sample storage for WGS samples.
- Overall turnaround times appear to have improved, however we are still awaiting further results to be able to show this. This is something we can re-visit 6 months after full implementation of all the changes. There have been additional improvements from the project which include the Genomic Tumour Advisory Board (GTAB) now happening fortnightly, instead of every month.
- Set up a generic email address to simplify access to CUP Whole Genome Sequencing (WGS). The result has enabled earlier diagnosis, and treatment of CUP Pan Tumour patient.
- Developed staff satisfaction surveys to understand and mitigate challenges in the CUP genomic pathway.
- WGS for patients with CUP is now available via the NHS genomic test directory. The professional survey found that staff feel the pathway is complicated but they are aware that is available on the NHS. However, they are either unaware of how to access it, or they have experienced issues with it in practice. The main issues focus on lack of standard pathway, lack of information, lack of digital tracking and recording of the request of samples. Delays at all parts of the pathway were highlighted. The final issues raised by the survey was lack of education and training. All the survey findings mirror the issues raised by the CUP WGS pathway process mapping, and are addressed in the process mapping action to aim to improve the pathway and make staff more aware of how to access the pathway and reduce delays.
- Trialling innovation tag tracking Radio Frequency Identification Device (RFID) system for tissue specimens to avoid additional cost and time implications and impact of the CWT.
- Detailed progress report on the CUP genomic pathway provided to the Outpatient Transformation Programme Board which has led to the development of a genetic form and communication plan to support easy access to genomic test results within the CCC Meditech system.
- There are still further planned improvements to be implemented including improved communication of the results to the CUP team from the Manchester team, electronic pathways from order to results, and tracking of samples.
- 28-day Cancer Waiting Time targets was not measured in this project as it was not relevant to the CUP pathway
Furthermore, the NHS and Clatterbridge Cancer Cancer (CCC) Strategy related to the outcomes is as follows:
- Be Collaborative: Collaborating with MSD, LCL/GLH, Mathew Krebs (NHSE), and the TRANSFORM project to improve the CUP pathway and reduce TAT.
- Be Outstanding: Faster diagnosis and access to cancer treatment, achievement of cancer targets (although CUP not within CWT).
- Be Research Leaders: Engaging in CtDNA research and working on CUP blood samples in advance of tissue samples.
- Be a Great Place To Work: Providing interim solutions for CUP genomic test results on the Meditech system, and providing access to CUP genomic education and resources on the CCC intranet.
MSD Benefits
- Better understanding of CUP Pan Tumour patient needs
- Enhance their reputation through partnership work
This collaboration may have enabled more patients to access innovative treatments in line with NICE guidance which may or may not have included MSD medicines.
Funding & Resources
This was a project with shared time commitment from Clatterbridge Cancer Centre & MSD
Patient Story2
Jane (pseudonym) is a 58-year-old female patient with Cancer of Unknown Primary (CUP).
She presented with dysphagia, vomiting and weight loss. After multiple investigations a CT scan showed a large posterior mediastinal mass, pleural and pericardial effusions. A biopsy showed an undifferentiated neoplasm of unknown origin. She was referred to the CUP MDT who agreed this was a cancer of unknown primary and care was taken over by the CUP team. A biopsy was taken for whole genomic sequencing (WGS) and she was started on standard chemotherapy, carboplatin and paclitaxel.
Jane told us:
“I didn’t really understand about the genomics tests, or why they might help, but after having a diagnosis of CUP, you would do anything to get more information about the cancer and you just hope it will help them (the team) have more understanding and more treatments”.
“I had almost forgotten about the genomic test when they called me. I had started chemo and was just focused on getting the treatment and fighting the cancer. When I came to clinic and explained the results, I didn’t really understand the details, but they told me the test had helped with finding a change in the cancer, but it also had signature that showed that I had a lung cancer, and they would refer me to the lung team for treatment moving forwards. This was a shock, but in a weird way I was pleased, it seemed like good news, I have a primary, and there were other treatments I could have”.
With the WGS findings Jane was referred for treatment under the lung team and had lung cancer specific chemotherapy.
Recent CT scan shows that the cancer is responding to the chemotherapy.
NHS Stakeholder quotes3,4:
“The collaboration has been excellent, working with MSD helped us process map the pathway and the outline issues, which we would have never done as well on our own. Also, regular project plan and clear actions helped me to account for the work I was supposed to be doing, to ensure timely actions and outcomes. Working with Wale has been fantastic, his support has meant we have made major service improvements, that on our own would not have happened this quickly.”
“Wale Ajet has been extremely knowledgeable, supportive and a pleasure to work with. He has ensured that the project remains on track but has done so without the small project group feeling under pressure. He ensures that every meeting is enjoyable and that is a skill set in itself. I should also pay credit to Nicola Harman who was instrumental in setting this project up.”
Lessons learnt
Maintaining patient privacy and confidentiality in the Cancer of Unknown Primary (CUP) pathway was crucial, given the small volume of patients involved. Ensuring appropriate consent was obtained and anonymising patient information were essential steps to protect privacy and confidentiality.
To increase the likelihood of implementing potential improvements identified through the output of the gap analysis, it was important to identify key stakeholders with decision-making responsibility. Engaging these stakeholders in the project helped translate identified improvements into actionable changes.
Outlining the roles and responsibility at each stage of the pathway, and then communicating clearly to all the stakeholders has been crucial to ensuring each step is correctly completed.
Ensuring visibility of the issues at multiple forums has helped engage stakeholders and make the improvements.
Engaging the digital team at an early point has been vital to making timely improvements.
There are still issues that are out with the gift of the project, and CCC, however, by demonstrating the issues at the correct forums this has helped provide evidence that change is needed.
Small team, expert knowledge, dedicated time, clear direction, expectations and shared vision. Knowledge of what is within gift to deliver and what is outside of scope – all of which were defined and therefore able to deliver to plan.
Publications
Genomic CQG
UK Acute Oncology Society Conference, Transforming Acute Oncology, is Everyone’s Business – September 28th Birmingham – Abstract to be submitted for a poster presentation.
References
- Clatterbridge Cancer Centre CUP 2024 patient tracker
- Feedback from cancer of unknown primary patient at The Clatterbridge Cancer Centre June 2024.
- MSD_Clatterbridge Cancer Centre CUP Pan Tumour PDP Workshop Survey
- MSD_Clatterbridge Cancer Centre CUP Pan Tumour PDP End of Project Survey
GB-NON-09803 | August 2024
Blackpool Teaching Hospitals NHS Foundation Trust Pan Tumour (Skin, Gynaecology, Upper GI) Diagnostic Pathology Pathway Development Project (PDP)
Project Title
Blackpool Teaching Hospitals NHS Foundation Trust Pan Tumour (Skin, Gynaecology, Upper GI) Diagnostic Pathology Pathway Development Project (PDP)
*Pan Tumour refers to the 3 tumour types been reviewed in this project Skin, Gynaecology & Upper GI
Organisations involved
MSD & Blackpool Teaching Hospitals NHS Foundation Trust
Summary
A national optimal Pan Tumour Diagnostic Pathology pathway has been created with the intention of improving patient experience through promoting quality cancer care and ensuring all Pan Tumour patients receive optimal cancer care. There is an opportunity in Blackpool to optimise the Pan Tumour Pathology pathway in line with the national optimal Pan Tumour Diagnostic Pathology pathway to improve the service quality, service efficiency, productivity and patient experience. The desired outcome of this project is an improved Pan Tumour Diagnostic Pathology pathway aligned with the national optimal pathway and achievement of the Pan Tumour Diagnostic Pathology pathway turnaround times reduction in line with the Royal College Pathology KPI (90% Sample received reported within 10 calendar days)1 and 28 day Faster Diagnostic Standard Cancer Waiting Time Targets. The project intends to run for approximately 6 months
Benefits Realised2
Through implementation of MSD’s Pathway Development Programme with Blackpool Teaching Hospital NHS Foundation Trust Histopathology team, MSD provided project management support to assess the turnaround times of their histopathology pathway from receipt of sample to diagnostic result reporting.
A gap analysis was carried out and the areas below were identified as area of improvement to reduce the turnaround times (TAT) reduction in line with the Royal College Pathology KPI (90% Sample received reported within 10 calendar days)1:
- Workforce – recruitment
- Increase Quality of Clinical information on request form.
- Weekend Working / extended working day.
- Skin diagnostic/skin treatment variation at triage to prioritise.
- Review rotas start times to cover full day use of H&E Stainer (particularly with additional staff now in lab).
- Competency assessment to ensure quality H&E image.
- Correct balance of staffing vs workload.
Patient Benefits:
Although patient experience was not directly measured as part of this project and any direct/indirect contact with patients were out of scope of this project, it is reasonably expected that the improved diagnostic pathway and reduced turnaround time would lead to quicker diagnoses and time to treatment. This could potentially result in improved patient outcomes. See NHS benefits below:
NHS Benefits:
The implementation of the suggested changes gradually reduced the diagnostic turnaround time in the histopathology laboratory despite high number of samples. With the September turnaround time showing for the first time all three-cancer type diagnostic and treatment samples reported below the 10-day turnaround time target. The improved laboratory process and new staff recruitment made a positive impact on the turnaround times.
As demonstrated below, the sample preparation was completed within 5-10 days in most cases and with a full consultant complement reported within 10 days target. However, due to the loss of two consultants in October and November, this negatively impact the progression seen.
3The graphs below highlight the phases of the project and the turnaround time (pre and post consultant) reporting achieved against the KPI of reporting 90% sample received within 10 days at each phase. Skin (TAT on average reduce from 14 to 12) GI (TAT on average reduce from 13 to 10), Gynae (TAT on average reduce from 11 to 10)

The project also aimed to achieve the 28-day cancer wait time target, however the data did not conclusively show a clear correlation between histopathology turnaround time and this target.
Earlier referral, diagnosis and treatment was not measured or used as a baseline.
Increase in treatment rates was not measured or used as a baseline as it was not relevant to histopathology diagnostic pathway.
MSD Benefits:
Through this project, MSD gained a better understanding of the histopathology diagnostic pathway and the needs of the NHS. The collaboration enhanced MSD’s reputation through partnership work. Additionally, the improved pathway may indirectly result in more appropriate usage of MSD’s NICE/SMC approved oncology medicines.
Quotes from NHS Stakeholders4
“The sessions we had supported by MSD were well planned and organised, the facilitators were well prepared with materials for the session and kept to time which was important. The sessions proved very informative and valuable for the team.”
“There were several improvements noted and implemented as a result of the work done with MSD that have made a difference to our service.”
This collaboration may have enabled more patients to access innovative treatments in line with NICE guidance which may or may not have included MSD medicines.
Funding & Resources
This was a project with shared time commitment from Blackpool Teaching Hospital NHS Foundation Trust, Lancashire Cancer Alliance & MSD
Lessons learnt
Several lessons were learned during the project, including the importance of sequential stakeholder engagement (have a series of touch point regularly) and clear communication plans, the need for a strong NHS project champion, and the impact of wider NHS workforce challenges on project delivery. It was recommended to engage project sponsors to ensure continuous prioritisation of the project throughout its duration.
The collaboration with non-promotional pharmaceutical implementation leads proved beneficial in supporting specific improvement work and utilising ‘free to the NHS’ additional capacity to benefit patients and providers. Kaye Davies, Lancashire South Cumbria Cancer Alliance (LSCCA) Programme Manager
Recommendation
Data revalidation and repeat audits around the turnaround time from receipt of sample to report on Skin, GI and Gynae diagnostic over 3, 6,9 month period separating diagnostic samples from treatment.
Publications
BTHT histopathology improvement presentation shared at LSCCA Cancer Board 23/24, LSCCA Stakeholder briefings 23/24
Planned to return to LSCCA Cancer Board 24/25 now complete and evaluated.
Sharing with secondary care stakeholders in Trusts via stakeholder briefings and cancer alliance bulletin
Sharing with Pathology Collaborative stakeholders via stakeholder briefings and cancer alliance bulletin
Reference:
- CP-WEB-INS-016.UN-16.1-Turnaround-times-V1.1-1
- Highlight report – Histopathology Improvement at BTH – BTH Histopathology Highlight report – Smartsheet.com.pdf
- Blackpool Histopathology Turnaround Time data – MSD Data (inc Date Sent).xlsx
- Sogolytics survey – Sogolytics – Omni Report
GB-NON-09632 | July 2024
Skin – Active Projects
Melanoma Pathway Development Programme (PDP) Swansea Bay University Health Board
Project Title
Melanoma Pathway Development Programme (PDP) Swansea Bay University Health Board
Organisations involved
Swansea Bay University Health Board and Merck Sharp and Dohme (UK) Limited
Summary
The intent of this collaboration is to optimise the melanoma patient pathway in Swansea Bay University Health Board (SBUHB). This will involve referral from dermatology to plastic surgery, through to treatment being given in oncology. The purpose of this is to improve the pathway service quality, efficiency, and productivity.
This will aim to support the achievement of the NHS Cancer Waiting Time (CWT) 62-day treatment target, as well as a reduction in time taken for subsequent treatment (post-excision). This has the potential to lead to improvements in patient experience of care. This PDP will be delivered collaboratively by both partners via the co-development and engagement in pathway mapping workshops and gap analysis to identify barriers and blocks.
Based on the findings from the gap analysis, both partners would subsequently work towards implementing plans of the identified areas of improvement and commit to measuring the outcomes of the project and disseminating these outcomes within 6 months of the project completion.
Background
There are complexities within the melanoma patient pathway within SBUHB, the pathway spans multiple sites across the two main hospitals in SBUHB, and after some initial discussions it is apparent there are variations and inefficiencies in this pathway.
There are some concerns that due to the complexities and variations of the pathway, some patients may not progress through the pathway in a timely manner, resulting in patients potentially missing out on treatment. There is therefore an opportunity in SBUHB to optimise the melanoma pathway to improve outcomes for melanoma patients.
Project Objectives
- Optimisation and standardisation of the melanoma patient pathway, reducing variation in cancer care across SBUHB.
- Understand the current patient pathway from the dermatology centres that feed into the specialist MDT, through to surgery and plastics and potential treatment in oncology.
- Understand the challenges, bottlenecks and inefficiencies within the patient pathway.
- Identify ways to address the challenges and obstacles within the patient pathway to optimise this pathway.
- Outline and reduce some of the inconsistencies seen in the existing patient pathway, reducing variation in care.
- Increase engagement and awareness of the melanoma patient pathway with dermatology, surgery, plastic surgery and oncology.
Benefits
Benefits/ Impact to patients:
- An improved patient experience of the melanoma pathway.
- Earlier referral and treatment of melanoma patients, improving the chance of successful treatment outcome.
Benefits to the NHS partner:
- Achievement of the skin cancer 62-day Cancer Wait Times.
- Earlier referral and treatment of skin cancer patients, improving the chance of successful treatment outcome.
- Increase in treatment rates for melanoma patients.
Benefits to MSD:
- Enhanced reputation of MSD through partnership work.
- As a pharmaceutical manufacturer of oncology medicines, an indirect result of an improved pathway may be that MSD see usage of their NICE approved medicines.
Funding & Resources
This project is a shared contribution of time between Swansea Bay University Health Board and MSD UK.
GB-NON-13011 | May 2026
NHS Grampian Melanoma Pathway Development Programme
Project Title
NHS Grampian Melanoma Pathway Development Programme
Organisations Involved
NHS Grampian and MSD
Summary
There is an opportunity in NHS Grampian to optimise the melanoma pathway to improve service quality, service efficiency, productivity and patient experience. The desired outcome of this project is to improve the efficiency of, or components of the melanoma pathway versus current baseline, including the achievement of the 62- and 31-day cancer waiting time targets that include time to surgery, radiotherapy and systemic anti-cancer therapy. The project is intended to run for approximately 12 months.
Background
The project is required to help address increasing pressure on the melanoma pathway, particularly in meeting the 62- and 31-day Cancer Waiting Times targets. Despite notable improvements in the quality of care and patient outcomes over the past decade, the 2022 Melanoma Cancer Quality Performance Indicators for Scotland report continues to highlight several areas needing attention across the pathway. Capacity constraints are causing delays in patient transit, further exacerbating pressure on services. Currently, consultants, nurses, and pharmacists are operating beyond their capacity, with demand expected to rise in the coming years, underscoring the urgent need for intervention.
Project Objectives
The objectives of the project are to establish a baseline understanding of the time taken to review melanoma patients by each specialty within the pathway, through a comprehensive review across multidisciplinary teams. It also aims to identify inefficiencies that are contributing to delays in patient transit, and to map the existing pathway in detail. This mapping will support a gap analysis, which NHS Grampian will use to explore targeted solutions that could reduce delays and improve outcomes for melanoma patients.
Benefits
Benefits to Patients
- An improved patient experience through the melanoma pathway in NHS Grampian.
- Potentially quicker time to treatment through improved pathway efficiencies.
Benefits to NHS Grampian
- Smoother and quicker referral process through the melanoma pathway from urgent suspected cancer referral to first treatment.
- Potential improvement of the 62-day and 31-day cancer waiting time targets.
- Potential efficiencies leading to more patients being treated in a faster time.
Benefits to MSD
- Better understanding of the patient journey through the melanoma pathway.
- Enhanced reputation of MSD through partnership work.
- As a pharmaceutical manufacturer of oncology medicines, an indirect result of an improved pathway may be that MSD see usage of their SMC reimbursed medicines.
Funding & Resources
This project is a shared contribution of time between NHS Grampian and MSD. No funding is associated with this collaborative working project.
GB-NON-11979 | October 2025
Melanoma Pathway Development Programme (PDP) Cwm Taf Morgannwg University Health Board
Project Title
Melanoma Pathway Development Programme (PDP) Cwm Taf Morgannwg University Health Board
Organisations involved
Cwm Taf Morgannwg University Health Board and Merck Sharp and Dohme (UK) Limited
Summary
The intent of this collaboration is to optimise the melanoma patient pathway in Princess of Wales Hospital (Cwm Taf Morgannwg University Health Board (CTMUHB)). This will involve referral from Dermatology to Plastic Surgery in Swansea Bay University Health Board (SBUHB) through to treatment being given in Oncology by Velindre University NHS Trust. The purpose of this is to improve the pathway service quality, efficiency, and productivity.
This will aim to ultimately support the achievement of the NHS Cancer Waiting Time (CWT) 62-day treatment target, as well as a reduction in time taken for subsequent treatment (post-excision). This has the potential to lead to improvements in patient experience of care. This would be delivered collaboratively by both partners via the co-development and engagement in pathway mapping workshops and gap analysis to identify barriers and blocks.
Based on the findings from the gap analysis, both partners would subsequently work towards implementing plans of the identified areas of improvement and commit to measuring the outcomes of the project and disseminating these outcomes within 6 months of the project completion.
Background
There are complexities within the melanoma patient pathway within CTMUHB, the pathway spans multiple sites (plastic surgery in SBUHB, Oncology in Velindre) and after some initial discussions it is apparent there are variations and inefficiencies in this pathway.
There are some concerns that due to the complexities and variations of the pathway, some patients may not progress through the pathway in a timely manner, resulting in patients potentially missing out on treatment. There is therefore an opportunity in CTMUHB to optimise the melanoma pathway to improve outcomes for melanoma patients.
Project Objectives
- Optimisation and standardisation of the melanoma patient pathway, reducing variation in cancer care across CTMUHB.
- Understand the current patient pathway from the dermatology centres that feed into the specialist MDT, through to treatment in Oncology.
- Understand the challenges, bottlenecks and inefficiencies within the patient pathway.
- Identify ways to address the challenges and obstacles within the patient pathway to optimise this pathway.
- Outline and reduce some of the inconsistencies seen in the existing patient pathway, reducing variation in care.
- Increase engagement and awareness of the melanoma patient pathway with dermatology, surgery, plastic surgery and oncology.
Benefits
Benefits/ Impact to patients:
- An improved patient experience of the melanoma pathway.
- Earlier referral and treatment of melanoma patients, improving the chance of successful treatment outcome.
Benefits to the NHS partner:
- Achievement of the skin cancer 62-day Cancer Wait Times.
- Earlier referral and treatment of skin cancer patients, improving the chance of successful treatment outcome.
- Increase in treatment rates for melanoma patients.
Benefits to MSD:
- Enhanced reputation of MSD through partnership work.
- As a pharmaceutical manufacturer of oncology medicines, an indirect result of an improved pathway may be that MSD see usage of their NICE approved medicines.
Funding & Resources
This project is a shared contribution of time between Cwm Taf Morgannwg University Health Board and MSD.
GB-NON-11931 | September 2025
Melanoma Pathway Development Programme (PDP) Portsmouth Hospitals University NHS Trust
Project Title
Melanoma Pathway Development Programme (PDP) Portsmouth Hospitals University NHS Trust
Organisations involved
PORTSMOUTH HOSPITALS UNIVERSITY NHS TRUST and MERCK SHARP & DOHME (UK) LIMITED
Summary
The intent of this collaboration is to optimise the Melanoma Patient Pathway in Portsmouth Hospitals University NHS Trust from referral from Dermatology to Surgical Oncology/Plastic Surgery through to treatment being given in Oncology at Queen Alexandra Hospital Portsmouth with the purpose of improving pathway service quality, efficiency and productivity.
This will aim to ultimately support the achievement of the NHS Cancer Waiting Time (CWT) 31-day treatment target, as well as a reduction in time taken for subsequent treatment (post-excision) and an increase in the number of patients receiving subsequent treatment. This has the potential to lead to improvements in patient experience of care. This will be delivered collaboratively by both partners via the co-development and engagement in pathway mapping workshops and gap analysis to identify barriers and blocks. Based on the findings from the gap analysis, both partners will subsequently work towards implementing plans of the identified areas of improvement and commit to measuring the outcomes of the project and disseminating these outcomes within 6 months of the project completion.
Background
There are complexities within the Melanoma patient pathway within Portsmouth Hospital NHS Trust; the pathway spans multi sites (Dermatology outside Queen Alexandra) and multi divisional units (Dermatology, General Surgery/Plastics & Oncology) and after some initial discussions it is apparent there are variations and inefficiencies in this pathway.
There are some concerns that due to the complexities and variations of the pathway, not all patients are being referred into Surgery/Plastics post local multidisciplinary team (MDT) through into the specialist MDT (SMDT) and onto Oncology in a timely manner (or at all), resulting in patients potentially missing out on treatment. Therefore, there is an opportunity in Portsmouth Hospitals University NHS Trust to optimise the melanoma pathway to improve outcomes for melanoma patients in Portsmouth.
Project Objectives
Optimisation and standardisation of melanoma patient pathway, reducing variation in cancer care across Hampshire by:
- Understanding the current patient pathway from the dermatology centres that feed into the specialist MDT, through to treatment in Oncology
- Understanding the challenges, bottlenecks and inefficiencies within the patient pathway
- Identifying ways to address the challenges and obstacles within the patient pathway
- Outlining and reducing any of the inconsistencies seen in the existing patient pathway, reducing variation in care
- Increasing engagement and awareness of the melanoma patient pathway with dermatology, surgery, plastic surgery and oncology
Benefits
Benefits/ Impact to patients:
- An improved patient experience of the melanoma pathway
- Earlier referral and treatment of melanoma patients, improving the chance of successful treatment outcome
Benefits to the NHS partner:
- Achievement of the skin cancer 31-day Cancer Wait Times
- Earlier referral and treatment of skin cancer patients, improving the chance of successful treatment outcome
- Increase in treatment rates for melanoma patients
Benefits to MSD:
- Enhanced reputation of MSD through partnership work
- As a pharmaceutical manufacturer of oncology medicines, an indirect result of an improved pathway may be that MSD see usage of their NICE approved medicines
Funding & Resources
This project is a shared contribution of time between Portsmouth Hospitals University NHS Trust and MSD
GB-NON-11662 | July 2025
South Yorkshire and Bassetlaw Cancer Alliance Skin Cancer Pathway Development Programme
Project Title
South Yorkshire and Bassetlaw Cancer Alliance Skin Cancer Pathway Development Programme
Organisations involved
South Yorkshire and Bassetlaw Cancer Alliance
Sheffield Teaching Hospital Foundation Trust
Rotherham NHS Foundation Trust
Barnsley NHS Foundation Trust
Doncaster and Bassetlaw Teaching Hospitals Trust
Chesterfield Hospital Foundation Trust
Summary
There is an opportunity across the South Yorkshire and Bassetlaw Cancer Alliance (SYBCA) region to optimise the skin cancer pathway in line with the national best practice timed skin pathway, to improve the service quality, service efficiency, productivity, and patient experience. The desired outcome of this project is an improved skin cancer pathway aligned with the national best practice timed pathway and achievement of the skin cancer 28 day faster diagnostic standard (FDS) and the 31- and 62-day cancer waiting time targets (CWT). The project intends to run for approximately 12 months.
Background
The aim of the pathway development programme (PDP) is to improve performance in this pathway by addressing pathway fragility and efficiency from clinical assessment (day 1) to decision to treat (DTT) (day 28) across the five trusts within this cancer alliance region (see organisations involved).
The project will primarily focus on the development of nurse led ‘see and treat’ models which is a cancer alliance deliverable for 2025. There will be alignment to the specialty mandate of the South Yorkshire and Bassetlaw Acute Federation, who have prioritised dermatology as part of their clinical services and sustainability review.
Project Objectives
- Pathway mapping to identify variation in skin cancer pathways.
- Prioritised list of improvement actions to deliver pathway standardisation, optimising use of dermatology and nurse led see and treat models of care.
- Improvement in CWT 28, 31 and 62 days and reduction in the percentage of breaches.
Benefits
Benefits to patients
- Improved patient experience of the skin cancer pathway.
Benefits to the NHS Partner
- Achievement of the skin cancer FDS 28 day, 31-day treatment target and 62-day referral to treatment CWT.
- Earlier referral, diagnosis and treatment of skin cancer patients, improving the chance of a successful treatment outcome.
- Workforce efficiencies realised.
Benefits to MSD
- Enhanced reputation of MSD through partnership work.
- As a pharmaceutical manufacturer of oncology medicines, an indirect result of an improved pathway may be that MSD see usage of their NICE/SMC approved medicines.
Funding & Resources
This project is a shared contribution of time between South Yorkshire and Bassetlaw Cancer Alliance and MSD.
GB-NON-11744 | August 2025
Skin – Completed Projects
University Hospitals Sussex Foundation Trust Melanoma Pathway Development Project (PDP)
Project Title
University Hospitals Sussex Foundation Trust Melanoma Pathway Development Project (PDP)
Organisations involved
- MSD-UK
- University Hospitals Sussex Foundation Trust
- Eastbourne District General Hospital & Conquest Hospital
- Worthing Hospital & St Richards Hospital
- Queen Victoria Hospital
- Royal Sussex County Hospital
Summary
The outcome summary for the University Hospitals Sussex Foundation Trust Melanoma Pathway Development Project (PDP) is currently pending due to finalisation and alignment between parties. We remain committed to publishing the full results once the required information is received and validated.
For background, a national optimal skin cancer pathway has been created with the intention of improving patient experience through promoting quality cancer care and ensuring all skin cancer patients receive optimal cancer care. There was an opportunity in Brighton and the surrounding Sussex area to optimise the melanoma pathway in line with the national optimal skin cancer pathway to improve the service quality, service efficiency, productivity and patient experience.
The purpose of this project was to optimise the Melanoma Patient Pathway from referral into Dermatology across the 4 skin MDT’s (Eastbourne District General Hospital & Conquest Hospital; Worthing Hospital & St Richards Hospital; Queen Victoria Hospital and Royal Sussex County Hospital) through to Specialist MDT(SMDT) at University Hospitals Sussex Foundation Trust (UHSx) and to treatment being given in Oncology at Royal Sussex County Hospital.
The desired outcome of this project was an improved melanoma pathway aligned with the national optimal pathway and achievement of the skin cancer 28 day Faster Diagnostic Standard and the 31- and 62-day Cancer Waiting Time Targets.
The Project began on 1st July 2024 and finished on 31st October 2025.
Next Steps
- Partner organisations will align on the data and associated outcomes.
- Once agreed, the outcome summary will be finalised and submitted for certification.
- We will update stakeholders promptly as progress is made.
Note:
This holding statement ensures transparency and compliance while we work collaboratively to complete the project deliverables.
Benefits Realised
Data is currently being collected, analysed and reviewed and will be updated in due course.
Publications
No publications are planned outside of this holding statement.
GB-NON-13012 | May 2026
University Hospitals Sussex Foundation Trust Melanoma Surveillance, Follow up and Discharge Pathway Development Programme (PDP)
Project Title
University Hospitals Sussex Foundation Trust Melanoma Surveillance, Follow up and Discharge Pathway Development Programme (PDP)
Organisations involved
- MSD-UK
- University Hospitals Sussex Foundation Trust
Summary
The outcome summary for the University Hospitals Sussex Foundation Trust Melanoma Surveillance, Follow up and Discharge Pathway Development Programme (PDP) is currently pending due to finalisation and alignment between parties. We remain committed to publishing the full results once the required information is received and validated.
For background, the demand within Oncology for melanoma has increased following the introduction of adjuvant treatment. Prior to adjuvant therapy, patients were discharged from oncology to dermatology or plastics with a clear standardised pathway in place. Currently there is no agreed standardised pathway for patients based on their stratified risk following their adjuvant treatment. Therefore, the current utilisation of dermatology, plastics and oncology appointments for follow-up are not optimised, in some cases all 3 specialties follow up these patients. Implementing an agreed standardised pathway for follow up following adjuvant treatment in line with the NICE Guidance (NG14) – Melanoma: Assessment and Management will ensure optimal usage of follow-up appointments within Oncology, Dermatology and Plastics.
Therefore, there was an opportunity in University Hospitals Sussex NHS Foundation Trust to implement and optimise patient pathways in melanoma following their adjuvant treatment. The aim of this project was to agree, document and implement new patient pathways for patients following their adjuvant therapy with Oncology, Dermatology and Plastics. This ensures there is a standardised approach for all patients following their adjuvant therapy to ensure their surveillance, follow-up, and discharge, if appropriate from Oncology, is consistent
The Project began on 1st August 2024 and finished on 30th September 2025.
Next Steps
- Partner organisations will align on the data and associated outcomes.
- Once agreed, the outcome summary will be finalised and submitted for certification.
- We will update stakeholders promptly as progress is made.
Note This holding statement ensures transparency and compliance while we work collaboratively to complete the project deliverables.
Benefits Realised
Data is currently being collected, analysed and reviewed and will be updated in due course.
Publications
No publications are planned outside of this holding statement.
GB-NON-12905 | May 2026
Writing of a business case for development of skin cancer services in Thames Valley
Project Title
Writing of a business case for development of skin cancer services in Thames Valley
Organisations Involved
Oxford University Hospitals NHS Foundation Trust (OUH) and MSD
Summary
The project aimed to address increasing patient demand, the introduction of adjuvant treatments approved by NICE, and the challenges in retaining staff at the OUH, all of which result in variation in treatment uptake, monitoring and ongoing surveillance. The objective was to provide project management support to understand the current state running of the service and write a business case for the permanent funding a senior band 7 cancer nurse specialist. This specialist would work to ensure seamless and coordinated care for all patients undergoing adjuvant therapy and surveillance monitoring within the skin cancer pathway at Oxford University Hospitals and all peripheral referring hospital sites. Once appointed the nurse will be able to provide care closer to the patient’s home, removing the need for travel and the likelihood of patients refusing adjuvant treatment. This will free up hospital consultant clinic slots for seeing all new patients, any patient with newly relapsed disease, patients on treatment for metastatic disease, and those patients with complications of treatment. At the time of writing, the OUH has agreed in principle to funding this new post and we are just waiting for the budget to be drawn down.
Benefits Realised
Benefits listed below include benefits realised as a result of the project as well as anticipated benefits once a senior band 7 cancer nurse specialist has been appointed. It has been stated where benefits listed are anticipated:
Patient Benefits (all are anticipated)
- Delivery of care closer to home – As the nurse will spread their work across Milton Keynes, Banbury, and Oxford – meeting one of the key tenets of the NHS Long Term Plan.1
- Increased accessibility for patient as clinics will be more accessible to individuals with limited mobility, those who rely on public transportation, or those who cannot afford the cost of travel. This will ensure that everyone, regardless of their socio-economic status, has better access to healthcare.
- Referral coordination if needed will ensure a seamless transition to surgical intervention or prompt systemic treatment for metastatic disease if the melanoma progresses.
- Seamless and coordinated care will potentially improve treatment outcomes.
- The nurse will provide emotional support and help to address any concerns that patients may have. This will ensure patients are able to express their anxieties and receive reassurance throughout the programme.
- Patient-focussed service with the creation of a more patient-centred and efficient active surveillance programme.
- Self-skin examination techniques and sun protection advice will empower patients to take an active role in managing their health.
NHS Benefits
- A business case has been written that justifies the need for the employment of a senior band 7 skin cancer nurse specialist who would provide seamless and coordinated care for all patients undergoing adjuvant therapy and surveillance monitoring within the skin cancer pathway across the region.
- Service data has been researched and included in the business case demonstrating:
- Increasing Patient Volumes: Significant growth in new and follow-up skin cancer patients, leading to immense pressure on consultants and service capacity.
- Clinical Risk: Inadequate follow-up appointments for surveillance, risking unobserved recurrence of skin cancer.
- Administrative Burden: High turnover of administrative staff due to constant rescheduling and booking challenges.
- Geographical Accessibility: Travel requirements for patients from Milton Keynes to Oxford, impacting patient acceptance of adjuvant therapy.
- Cost and Efficiency: Financial benefits of employing a nurse specialist, freeing up consultant time and improving service efficiency.
NHS Benefits (anticipated)
- The new nurse will be able to deliver patients’ appointments closer to their homes in Milton Keynes and Banbury, reduce the admin burden associated with ‘further reach’ patients travelling into Oxford, and improving patient experience.
- Consultants will have clinic slots freed up for seeing all new patients, any patient with newly relapsed disease, patients on treatment for metastatic disease, and those patients with complications of treatment.
- By seeing all the follow-up patients, the new nurse would release 846 hours of consultant time every year (time cost equivalent to £61,512 of consultant time released each year).
- £15.23 will be saved for every follow-up patient seen by a skin cancer nurse rather than the consultant.2
- Future service resilience will be created to meet the anticipated >10% annual growth rate in patient demand (12% anticipated average growth in new patients and ~12% anticipated growth in ‘follow-up’ patients).3
MSD Benefits
- Once the nurse has been appointed and operating fully, we believe that 1835 patients will be positively impacted each year. This has been calculated using NHSE cancer waiting times data.4
- An opportunity to contribute towards the development of cancer services in the skin pathway, gaining experience of how to create a business case that draws attention to under-resourced areas of the pathway.
- Collaborating on a project that has the potential to positively impact patients through a business case that may be applicable to other geographies and speaks to the NHSE cancer workforce agenda.5
- This collaboration may ultimately have enabled more patients to access innovative treatments in line with NICE guidance which may or may not have included MSD medicines.
Funding & Resources
This was a project with shared time commitment from Oxford University Hospitals & MSD.
Lessons Learnt
- It is worth investing in a business case should the justification be strong enough, even when no recognised funding stream is apparent in the first instance. This is because writing a business case formalises the requirement and quantifies and qualifies the need in a formal document that is then more likely to be acted upon.
- Speaking to a broad range of clinical stakeholders enables a more diverse perspective to be included in the case for change.
- Uptake of adjuvant therapy has increased the need for senior skin cancer nurse specialists to be appointed, whose remit is adjuvant patients and those in surveillance programme.
Publications
None
References
- The NHS in England at 75 (NHS@75) – Priorities for the Future (2023)
- Figures calculated by subtracting top tier band 7 nurse salary from consultant salary (with 8 years completed service)
- Data obtained from OUH service data analysis measuring skin cancer patient demand changes year on year over the last 6 years
- NHSE Cancer Waiting Times Data, total skin cancer patients seen in the 31-day pathway over a 12-month period from Jan to Dec 2024
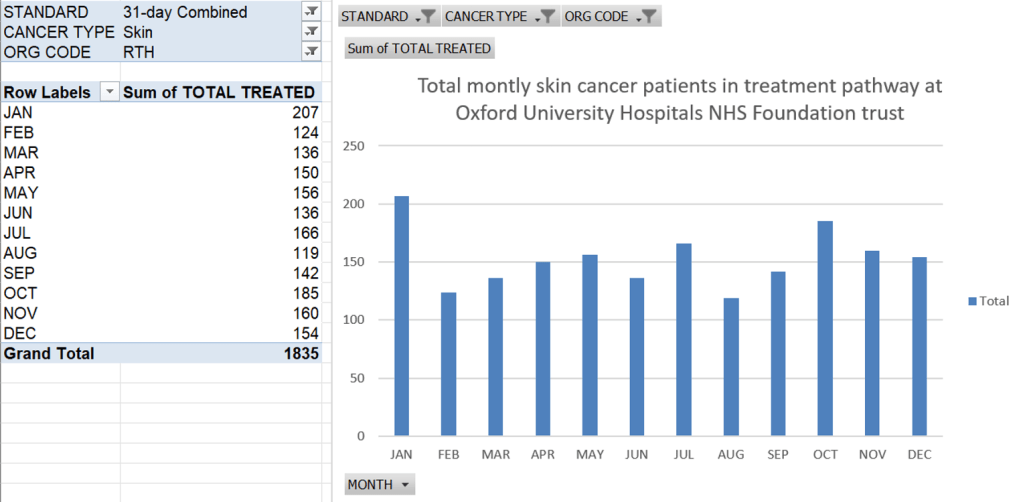
GB-NON-11077 | March 2025
Maidstone & Tunbridge Wells NHS Trust Skin Cancer Pathway Development Project (PDP)
Project Title
Maidstone & Tunbridge Wells NHS Trust Skin Cancer Pathway Development Project (PDP).
Organisations Involved
MSD.
Maidstone & Tunbridge Wells NHS Trust.
Summary
There is an opportunity in Maidstone & Tunbridge Wells to optimise the skin cancer pathway to improve the service quality, service efficiency, productivity, and patient experience. The desired outcome of this project is an improved skin cancer pathway and achievement of the skin cancer 28 day Faster Diagnostic Standard and the 31- and 62-day Cancer Waiting Time Targets. The project ran from June 2023 to June 2024.
Benefits Realised
Through implementation of MSD’s Pathway Development Programme with Maidstone & Tunbridge Wells NHS Trust, MSD provided project management support to assess the current state of the Incidental Findings of Melanoma Pathway and provide a gap analysis workshop to identify areas for improvement. MSD then supported the implementation of improvement initiatives to close these gaps and assisted with the measurement of their impact. As a result of the project, the following benefits were realised:
Patient Benefits
- Quicker diagnosis and treatment of Skin Cancer and hence improving the chance of successful treatment has been reported through this project potentially leading to improved patient outcomes, see NHS benefits below.
- Patient experience was not measured as part of this project.
NHS Benefits
An optimised pathway in skin cancer across Maidstone & Tunbridge Wells hospital sites resulting in:
- Optimisation of service delivery – Implementation of an updated regional referral pathway for incidental findings of Melanoma into specialist MDT.
“By way of feedback to you Dr X has already used the new referral form and it has worked very well – certainly better than his last experience” Lead Pathway Nurse Coordinator, General Surgery.
- Skin Cancer 28 day Faster Diagnostic Standard- was not measured as part of this project and was out of scope as patients are already diagnosed before entering the incidental pathway.
- Skin Cancer 31-day Treatment Cancer Wait Time Target increased from 86.4% June 2023 to 100% June 2024.
- Skin Cancer 62-day Referral to Treatment Cancer Wait Time Target- was not measured as part of this project as not reflective of the incidental findings pathway.
- Earlier referral, diagnosis and treatment of skin cancer patients as demonstrated by:
- Decision to treat to subsequent treatment (Oncological) decreased by 3 days average from 20 days (Feb-June 2023) to 17 days (June-Oct 2024).
- Increase in treatment rates for skin cancer – Number of patients treated by Oncology (First definitive, subsequent & recurrence) increased from 9 per month on average (Mar-June 2023) to 10 per month on average (June-Sept 2024).
MSD Benefits
- Better understanding of skin cancer patient needs and the need for rapid referral into specialist MDT to enable earlier treatment decisions.
- Enhanced reputation of MSD through partnership work.
- This collaboration may have enabled more patients to access innovative treatments in line with NICE guidance which may or may not have included MSD medicines.
Funding & Resources
This was a project with shared time commitment from Maidstone & Tunbridge Wells NHS Trust & MSD.
Lessons Learnt
- From the outset understanding deliverables required for optimisation of a pathway and the data required to demonstrate the success of the deliverables. We experienced challenges extracting the data required from hospital IT systems. Ensuring data services are bought in at the scoping phase of project to establish what can or can’t be measured.
- Ensure regular touch points throughout the project to ensure clarity of expectations and outcomes.
- Communication is key between all parties which is especially important between departments to optimise the decision making and establish the most appropriate intervention for the patient.
Publications
There are currently no plans to publish the outcomes of this project by the hospital trust as per the date of this summary.
GB-NON-10665 | November 2024
Vaccines Collaborative Working
Vaccines – Active Projects
NHS Human Papillomavirus (HPV) Immunisation Catch-Up Across North West Leicestershire GP Federation
Project Title
NHS Human Papillomavirus (HPV) Immunisation Catch-Up Across North West Leicestershire GP Federation.
Organisations involved
North West Leicestershire (NWL) GP Federation
MSD UK Ltd
Summary
This project aims to perform patient record searches and recall activity provided by NWL GP Federation. This will enable the updating of patient records, those identified as unvaccinated and in the catch-up cohort will be offered the HPV vaccination via extra clinics delivered across North West Leicestershire and using video invitations to call and recall patients.
The project will run for 9 months, and the expected outcome is an improvement in access to the NHS HPV immunisation programme in the HPV catch-up cohort in the North West Leicestershire area.
Background
Throughout the COVID-19 pandemic, we saw vaccine coverage rates (for non-COVID immunisation programmes) dramatically decrease across the UK. The new NHS Vaccination Strategy details tangible measures to empower the NHS, at every level, to maximise protection against vaccine-preventable diseases.
Preliminary data also suggests there has been a constant decline in the childhood immunisation uptake over 10 years (2014 to 2024). Since the COVID-19 pandemic, a noticeable decline in uptake has been observed across all immunisation programmes including childhood, adolescent, and adult vaccination and immunisation programmes.
The Department of Health and Social Care has already centrally procured HPV vaccination via a negotiated agreement; access to the vaccine is free in the UK up until the age of 25 via a National Immunisation Programme (NIP) that is delivered by NHS England through a school immunisation programme. Given there has been a decline in vaccination coverage rates (VCRs) there is a need for public health reasons to increase VCRs.
Project Objectives
- Improving access to NHS HPV immunisation programme in the HPV catch-up cohort.
- Delivering healthcare in an equitable way by offering out of hours clinics across varying socioeconomic areas.
- Testing the effectiveness of innovative ways of engaging with patients by using video invitations for call/recall.
- Improving quality of HPV vaccination data in Primary Care.
- New GP Federation HPV vaccination delivery model developed for longer term use beyond end of the Collaborative Working project.
Benefits
Patients
- Potential to improve access to NHS HPV immunisation programme (out of hours access) for those people in the HPV Catch-up cohort
NHS
- Support to run out of hours clinics alleviating practice capacity issues
- Public health impact if more people vaccinated against preventable disease
- Opportunity to gauge effectiveness of video invitations
- Blueprint to implement in other geographical areas
- Improved accuracy in patient records of HPV vaccine status
- Potential to provide evidence for future funding from commissioners
MSD
- Enhanced reputation and demonstration of commitment of MSD through partnership work in vaccines
- The learnings can be used to inform future projects with the patient at the centre of everything we do
- Successful deployment could be scaled to other vaccines, and shared with other areas
Funding & Resources
This project is a shared contribution between North West Leicestershire GP Federation and MSD. The total project cost is £21,250
GB-NON-12899 | April 2026
NHS National Human Papillomavirus (HPV) Immunisation catch-up programme and Cervical Screening programme awareness across North Central London Integrated Care Board
Project Title
NHS National Human Papillomavirus (HPV) Immunisation catch-up programme and Cervical Screening programme awareness across North Central London Integrated Care Board
Organisations involved
North Central London Integrated Care Board (NCL ICB)
Summary
The aim of this project is to provide enhanced access to HPV vaccination for young adults aged 14 and over, and less than 25 years old who have not had their vaccination within North Central London. This will be done via extra clinics, led by NCL ICB, supported by their member GP practices.
Within North Central London, an initial record search has identified 55% of males & females aged 14 and over, and less than 25 years old, have had HPV vaccination, indicating 45% have not.
This project aims to interrogate the initial data via secondary patient record searches and recall activity provided by NCL ICB. This will enable the updating of patient records, those identified as unvaccinated will be offered the HPV vaccination via GP member practices across North Central London.
The project will also seek to educate and inform about the importance of taking up the National Health Service’s existing offer of vaccination and cervical cancer screening when offered it. The project will run for 12 months and the expected outcome is an improvement in access to the NHS National Immunisation Programme for young adults aged 14 and over and less than 25 years old. As well as an improvement in cervical screening uptake in women aged 25 years and above.
Background
Throughout the COVID-19 pandemic, we saw vaccine coverage rates (for non-COVID immunisation programmes) dramatically decrease across the UK as health inequalities were starkly exposed. The new NHS Vaccination Strategy details tangible measures to empower the NHS, at every level, to maximise protection against vaccine-preventable diseases.
Preliminary data also suggests there has been a constant decline in the childhood immunisation uptake over the last 10 years (2014 to 2024). Since the COVID-19 pandemic, a noticeable decline in uptake has been observed across all immunisation programmes including childhood, adolescent, and adult vaccination and immunisation programmes.
The Department of Health and Social Care has already centrally procured the HPV vaccine via a negotiated agreement; access to the vaccine is free in the UK up until the age of 25 via a National Immunisation Programme (NIP) that is delivered by NHSE through a school immunisation programme. Given there has been a decline in vaccination coverage rates (VCRs) there is a need for public health reasons, and to avoid medicine wastage, to increase VCRs.
Project Objectives
- Data validated by second searches run by NCL ICB GP practices, these GP practices will also deliver call/recall activity
- Patient records updated, improving the accuracy of HPV status for local public health reporting purposes
- Gain a better understanding of vaccine and screening hesitancy
- Gather insight into the best communication methods and materials to contact and inform this age group about health matters
- Work with the Cancer Alliance to increase awareness of the pathway to eliminate cervical cancer, from vaccination to cervical screening, and early treatment.
Benefits
Patients
- Potential to improve access to the NHS National Immunisation Programme in young adults aged 16 and over, and less than 25 years old
- Improving access to cervical screening to women 25 years and older, due to raising awareness and education regarding the importance of screening
- Reducing the risk of cancers related to the human papillomavirus
NHS
- Reducing inequalities in North Central London
- Reducing the risk of cancers related to the human papillomavirus
- Improved accuracy in patient records
- Potential to provide evidence for future funding from commissioners
- Utilising already procured vaccine stock so reducing wastage and NHS disposal and storage costs
MSD
- Enhanced reputation and demonstration of commitment of MSD through partnership work in vaccines
- The learnings can be used to inform future projects with the patient at the centre of everything we do
- Successful deployment could be scaled to other vaccination programmes, and shared with other areas
Funding & Resources
This project is a shared contribution between North Central London Integrated Care Board and MSD. The total project cost is £182,305.
GB-NON-12385 | December 2025
NHS National Human Papillomavirus (HPV) Immunisation catch-up programme across Mid-Hampshire
Project Title
NHS National Human Papillomavirus (HPV) Immunisation catch-up programme across Mid-Hampshire
Organisations involved
Mid-Hampshire Healthcare Limited
Summary
The aim of this project is to provide enhanced access to HPV vaccination for young adults aged 16 and over, and less than 25 years old who have not had their vaccination within Mid-Hampshire Healthcare Ltd GP Federation. This will be done via extra clinics and outreach, led by Mid Hampshire Healthcare Ltd.
Within Winchester and Andover, an initial record search has identified 3,600 males & females aged 16 and over, and less than 25 years old, who are unvaccinated against HPV.
This project aims to interrogate the initial data via secondary patient record searches and recall activity provided by Mid-Hampshire Ltd. This will enable the updating of patient records, those identified as unvaccinated will be offered HPV vaccination via extra clinics and outreach across Mid-Hampshire.
The project will run for 6 months, and the expected outcome is an improvement in access to the NHS National Immunisation Programme for young adults aged 16 and over, and less than 25 years old.
Background
Throughout the COVID-19 pandemic, we saw vaccine coverage rates (for non-COVID immunisation programmes) dramatically decrease across the UK as health inequalities were starkly exposed. The new NHS Vaccination Strategy details tangible measures to empower the NHS, at every level, to maximise protection against vaccine-preventable diseases.
Preliminary data also suggests there has been a constant decline in the childhood immunisation uptake over the last 10 years (2014 to 2024). Since the COVID-19 pandemic, a noticeable decline in uptake has been observed across all immunisation programmes including childhood, adolescent, and adult vaccination and immunisation programmes.
The Department of Health and Social Care has already centrally procured HPV vaccination via a negotiated agreement; access to the vaccine is free in the UK up until the age of 25 via a National Immunisation Programme (NIP) that is delivered by NHSE through a school immunisation programme. Given there has been a decline in vaccination coverage rates (VCRs) there is a need for public health reasons, and to avoid medicine wastage, to increase VCRs.
Project Objectives
- Validate member practice data against CHIS data
- Data validated by second searches being run by Mid Hampshire Healthcare (MHH) Ltd with data sharing agreements in place with GP practices, MHH will also deliver recall activity
- Patient records updated, improving the accuracy of HPV status for local public health reporting purposes
- New GP Federation HPV vaccination delivery model developed for longer term use beyond end of the Collaborative Working project
Benefits
Patients
- Potential to improve access to the NHS National Immunisation Programme in young adults aged 16 and over, and less than 25 years old
NHS
- Reducing inequalities in specified localities within Mid-Hampshire
- Improved accuracy in patient records
- Potential to provide evidence for future funding from commissioners
- Utilising already procured vaccine stock so reducing wastage and NHS disposal and storage costs
MSD
- Enhanced reputation and demonstration of commitment of MSD through partnership work in vaccines
- The learnings can be used to inform future projects with the patient at the centre of everything we do
- Successful deployment could be scaled to other vaccines, and shared with other areas
Funding & Resources
This project is a shared contribution between Mid-Hampshire Healthcare Ltd and MSD. The total project cost is £25,000.
GB-NON-12386 | December 2025
Vaccines – Completed Projects
Developing a new solution for the delivery of vaccination and immunisations in Nottingham and Nottinghamshire, United Kingdom
Project Title
Developing a new solution for the delivery of vaccination and immunisations in Nottingham and Nottinghamshire, United Kingdom
Organisations involved
MSD UK Ltd and Nottingham & Nottinghamshire Integrated Care Board and NHS Confederation
Summary
The aim of the project was to co-design a vaccine delivery solution that was: (a) responsive to local communities; (b) provides effective local delivery; and (c) solves the specific challenges of low uptake of vaccinations and immunisations in Nottingham and Nottinghamshire. NHS Confederation’s Five Step Process was used to critically assess the data which informed potential solutions.
Given the size and complexities of the various cohorts concerned, and to fit within the planned 6-month timescale, the project focussed specifically on Flu and Covid vaccinations, in the Nottingham City and Nottingham North areas of the ICB, to include under 65s, pregnant women, people who are ‘at risk,’ and those with long term conditions as identified in the JCVI recommendations for the respective programmes.
The project ran for 6 months from August 2025 – January 2026 and the outcome is a new model of delivery to improve Flu and Covid vaccination accessibility and uptake that is tailored to local communities.
Benefits Realised
The new model enables a move from a reactive, provider-by-provider approach model to a neighbourhood-led Vaccination Delivery Network that treats primary care and community pharmacy as one coordinated system. The intention and longer-term benefit is to improve uptake (especially in under-vaccinated cohorts), to reduce duplication and wastage, and to stabilise provider participation as programmes expand.
Patients
- Maximising Voluntary Care Sector organisations with established relationships with population cohorts of low uptake, the aim is to provide greater access to information to allow individuals to make more informed decisions. Application of the RSPH ‘Pathway of Trust’ – https://www.rsph.org.uk/our-work/publications/building-a-path-to-trust-promoting-vaccinations-in-a-way-that-serves-communities/
- Improved booking, access and patient journey: (i) consistent invitations (call/recall); (ii) clear pharmacy walk-in/same-day offer for convenience cohorts; (iii) targeted neighbourhood vaccination events for equity cohorts at trusted venues/times
- Reducing inequalities: (i) place-based equity micro-plans for Black African/Caribbean, South Asian, and Eastern European communities; (ii) community-led messaging (trusted voices) using the 3Cs: confidence, complacency, convenience
NHS
- Deployment of enabling activities and improved Governance: (i) formal GP–pharmacy collaboration at PCN/Place: shared cohort allocation, capacity planning, pooled wastage risk; (ii) standardised booking/invitations and shared reporting; (iii) system Vaccination Delivery Board with Place/PCN operational groups, and LMC/LPC input
- Improved Data & IT Connectivity: (i) year 1 non-negotiable: minimum viable data standard (coding guidance, reconciliation, interoperability roadmap); (ii) quick wins: coding guidance, shared dashboards; 2–3-year digital roadmap with ICS digital leadership
- Workforce & Skill Mix: (i) joint workforce plan across GP, pharmacy, community services; surge staffing and shared standards; (ii) build flexible options: mobile teams, PCN hubs, bank/agency staff as needed
- Vaccine Ordering, Wastage & Financial Risk (Risk Share): (i) pilot pooled wastage risk at PCN/Place to reduce defensive ordering and stabilise participation (ii) define outcome-based incentives (e.g., uptake thresholds in priority cohorts) with clear guard rails
- Deploy a resilient, scalable operating model for new vaccines and eligibility changes
MSD
- Enhanced reputation and demonstration of commitment of MSD through partnership work in vaccines
- The learnings can be used to inform future projects with the patient at the centre of everything we do
- Successful deployment of the new model of delivery could be scaled to other vaccines, some of which are manufactured by MSD
- This collaboration may enable more patients to access innovative treatments and vaccinations in line with NICE & JCVI guidance which may or may not have included MSD medicines.
Funding & Resources
This project is a shared contribution between Nottingham & Nottinghamshire Integrated Care Board and MSD. The total project cost is £35,000.
Lessons learnt
- A new system model is a complex project requiring support from an external partner
- A ‘one size fits all’ access models do not meet population needs
- Community pharmacy can be underutilised, and capable of combining with primary care resources to improve access and uptake of vaccinations through addressing confidence, convenience, and complacency barriers
- Consider external environment impact (major system changes taking place in parallel with project delivery), together with post pandemic elective care pressures and impact from Industrial action
- It’s critical to base solutions on intelligence gathered rather than initial assumptions
- Be realistic on project timeframe and what can be implemented in the 6 months
- Related to the above, be clear it is setting goals to achieve by short, medium and long term
- Equity requires targeted neighbourhood-led action
Publications
- NHS Confed Best practice sharing webpage -. Ways of Working (in next phase of implementation to include certification by MSD)
- MSD Engage
Job code: GB-NON-12816 | April 2026
Read about our range of partnerships through some of our stories:
[Hosting Page GB-NON-12323 | Nov 25]



